
An updated narrative review of treatment for limbal epithelial stem cell deficiency
Introduction
Background
Limbal stem cell deficiency (LSCD) is a rare disease. Its treatment is often difficult. LSCD is a condition in which normal corneal epithelization is not maintained because of the loss of limbal stem cells (LSCs) (1,2). Diseased corneal surface is covered by opaque conjunctiva with neovascularization. In severe conditions involving the visual axis, visual rehabilitation should be achieved using stepwise surgical approaches including multiple surgeries such as LSC transplantation and corneal transplantation.
Rationale and knowledge gap
Since the pioneering works by Kenyon and Tseng who first proposed the role of LSCs in corneal epithelial homeostasis in 1989, many basic and clinical studies have been reported in this field (1). Various cell markers for LSCs have been found and innovative surgical treatment modalities have been developed (3). Recently, advanced stem cell technology has been incorporated into the development of a novel cell therapy to replace LSCs.
Objective
The aim of this review was to summarize basic knowledge of LSCD as clinicians and a general concept of its diagnosis and treatment. This article is presented in accordance with the Narrative Review reporting checklist (available at https://aes.amegroups.com/article/view/10.21037/aes-22-51/rc).
Methods
A PubMed literature review was performed to search for relevant articles on LSCs and LSCD published in English from May 1989 to May 2022. The search strategy is presented in Table 1.
Table 1
| Items | Specification |
|---|---|
| Date of search | July 25th, 2022 |
| Databases and other sources searched | PubMed |
| Search terms used | Limbal, limbal stem cell, keratoprosthesis, limbal deficiency |
| Timeframe | May 1989 – May 2022 |
| Inclusion and exclusion criteria | Inclusion: English publication, discusses limbal stem cell and limbal stem cell deficiency, partial LSCD, total LSCD, animal models, clinical studies, case Exclusion: non-English publication; not discussing limbal stem cell and limbal stem cell deficiency |
| Selection process | The author reviewed the papers involved and agreed to review them based upon the relevance of the articles |
LSCD, limbal stem cell deficiency.
Discussion
LSCs
It has been known for a long time that stem cells for regenerating corneal epithelium exist. The exact location of these stem cells was found to be the limbus about 30 years ago. Using radio-labeling technique, Cotsarelis et al. (4) have verified that there are label-retaining, slow-cycling, and stem like cells at the limbus. These cells progressively lost radio-labeling as they migrated toward the central cornea.
LSCs reside in the limbal stem cell niche, a unique environment at the junction between the corneal epithelium and the conjunctival epithelium. The limbal stem cell niche has the classical architecture of the palisade of Vogt, limbal stem cell crypt, and limbal crypt or focal stromal projection near the palisade of Vogt (5). These structures are more prevalent in the superior and inferior limbus. The niche provides protection from ultraviolet light and supplies LSCs with blood vessels and various signaling molecules. Recently, there were some evidences in animals and human to support the existence of LSCs even at the central cornea (6-8). However, it is still an established theory that most LSCs reside in the limbal area.
Molecular markers of LSCs are important for isolating and harvesting LSCs and culturing them for transplantation. For a long time, many researchers have tried to find molecular markers to characterize LSCs. However, there are no established markers for LSCs yet. At present, cytokeratins (such as cytokeratin K15), ΔNp63α, C/EBPσ, Bmi1, ABCG2, and Notch-1 are well known candidate markers for LSCs (9).
Limbal stem cell niche
The LSC niche is located in the Palisades of Vogt that are radially oriented, usually pigmented, and more prominent in superior and inferior limbi. Unlike the cornea, basement membrane at Palisades of Vogt is undulating with papillae, crypts, or stromal projections and fenestrated. This unique structure can shelter LSCs from physical shearing stress and provide a larger surface area to accommodate more LSCs within the confined area. Pigments of surrounding melanocyte can protect LSCs from ultraviolet damage (10). Limbal stroma underlying the Palisades of Vogt is heavily innervated and vascularized.
The interaction of stem cell with the surrounding environment such as vessel, neurons, and extracellular matrix is important in the homeostasis or activation of stem cells. How LSCs divide and maintain homeostasis has not been clarified yet. However, many studies have speculated that the unique three-dimensional structure of the limbus might play a critical role. Characteristically, the basement membrane at the Palisades of Vogt expresses laminin α2β2 chains, whereas the corneal basement membrane does not. In addition, α1, α2, and α5 chains of type IV collagen are present in the limbal basement membrane, while α3 and α5 chains are present in the central cornea. These properties may assist in sequestering and modulating concentrations of growth factors and cytokines for efficient and precise targeting to LSCs (11).
Etiology of LSCD
LSCD can be acquired or congenital. In most cases, it is acquired (12). The most common etiologies of LSCD are chemical/thermal burns, allergic conjunctivitis, Stevens Johnson syndrome, and mucous membrane pemphigoid, whereas the most common etiology of congenital LSCD is aniridia (2). Common etiologies of LSCD are described in Table 2.
Table 2
| Primary causes |
| Aniridia |
| Peter’s anomaly |
| Ectrodactyly-ectodermal-dysplasia-clefting syndrome |
| Xeroderma pigmentosum |
| Turner syndrome |
| Dyskeratosis congenita |
| Multiple endocrine deficiency |
| Congenital erythrokeratodermia |
| Secondary causes |
| Thermal or chemical burn |
| Excessive contact lens wear |
| Ocular inflammation |
| Stevens-Johnson syndrome |
| Toxic epidermal necrolysis |
| Ocular cicatricial pemphigoid |
| Atopic keratoconjunctivitis |
| Herpes keratitis |
| Trachoma |
| Neurotrophic keratitis |
| Radiation therapy |
| Iatrogenic |
| Mitomycin C |
| 5-FU |
| Cryotherapy |
| Extensive pterygium |
5-FU, 5-fluorouracil.
Symptoms and signs of LSCD
In its early stages, LSCD is usually asymptomatic. When LSCD progresses, various symptoms can occur, including blurring, ocular discomfort, photophobia, tearing, conjunctiva injection, and pain (3). Patients usually experience these symptoms months to years before diagnosis.
Typical early sign of LSCD is extension of the conjunctival epithelium (slight loss of transparency of epithelium) with fine vessels crossing the anatomic limbus. This is more common in the superior limbus. Punctate epithelial staining, tear film dysfunction, and conjunctival injection can occur when the disease progresses. At the late stage, corneal vascularization (superficial and deep) and fibrovascular pannus can develop and lead to visual loss.
Diagnosis of LSCD
The diagnosis of LSCD is primarily based on slit-lamp findings. An irregular corneal epithelium and conjunctivalization with neovascularization of the involved area suggest LSCD. Close observation of the corneal limbus may reveal disappearance of the palisade of Vogt. Fluorescein staining highlights the irregular, whorl-like pattern of the corneal epithelium (5). Sometimes, the demarcation line between the cornea epithelium and the invading conjunctival epithelium can be observed (12). Persistent epithelial defect is another important sign of LSCD. When the disease progresses further, corneal melting and perforation can occur.
Various objective diagnostic tools for LSCD are available. Impression cytology can reveal the presence of goblet cells in the conjunctivalized area (12). However, the absence of goblet cells does not necessarily imply intact LSCs. Additional immunocytochemistry is useful to find cells expressing conjunctival epithelial markers (such as cytokeratins 7, 13, and 19) or goblet cell markers such as mucin 5AC (13,14). Cytokeratin 12 is a corneal epithelial cell marker (15). Goblet cells and decreased density of the sub-basal nerve plexus can be detected in patients with LSCD using non-invasive in vivo confocal microscopy (16). LSCD can induce subtle changes in cornea epithelial layers, such as thinning of the epithelial layer or increased reflectivity of the epithelium. These changes can be imaged using anterior segment optical coherence tomography (OCT) (17). OCT angiography can also detect limbal vascular changes in patients with LSCD (18).
Medical treatment of LSCD
Except for sudden trauma such as chemical burn, diseases causing LSCD can slowly deplete LSCs. The damage is further promoted by uncontrolled inflammation, unstable tear film, and eyelid abnormalities. Therefore, treatment is needed to minimize LSC damage at an early stage of the disease to prevent further compromise of residual LSCs. Correction of the eyelid and tear film abnormalities with effective control of ocular surface inflammation may prevent the progression of LSCD to symptomatic stages.
Growth factor therapy is also effective in protecting the remaining LSCs in the limbal niche (3). Autologous serum, platelet-rich plasma, and amniotic membrane extract eyedrops contain various growth factors that can revitalize the limbal niche have the potential to reverse LSCD at an early stage (19). However, these treatments have limited efficacy for LSCD at its advanced stages.
Surgical procedures
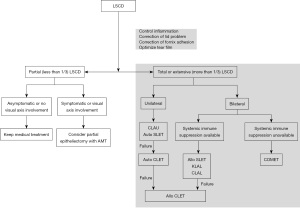
Mild degree of partial LSCD can be treated by simply removing abnormal epithelium and allowing the denuded cornea to be resurfaced with cells from the remaining intact LSCs. However, if LSCD has progressed with invasion of the visual axis, advanced surgical treatment is required. The goal of surgery is to transfer a sufficient amount of LSCs or corneal epithelial-like cells to the diseased ocular surface to promote and maintain corneal epithelialization. Although many types of limbal epithelial cell transplantation technique have been developed, none of them is universally successful.
Among various surgical methods introduced, the choice of surgical methods should be based on a thorough consultation between the patient and the surgeon. Depending on the surgical method, the patient may be at risk of damage to the healthy contralateral eye or long-term systemic immunosuppression if an allograft is used. Harvesting LSCs from the contralateral eye (e.g., conjunctival limbal autograft, CLAU) can induce iatrogenic LSCD in the donor eye (20). To avoid the risk of iatrogenic LSCD or bilateral advanced LSCD where enough autologous LSCs cannot be harvested, cell culture methods such as autologous cultivated limbal epithelial transplantation (CLET) can be used (21). Alternatively, simple limbal epithelial transplantation (SLET) recently introduced that requires only the use of 2×2 mm-sized small limbal block from the healthy donor eye can be used to minimize the risk of iatrogenic LSCD (22).
SLET deserves further discussion. Other surgical methods developed so far can transplant the harvested graft onto the limbus, the primary area of disease. However, in SLET, harvested limbal graft is cut into 6–10 pieces. These pieces are placed in the mid-periphery of cornea in a concentric pattern using fibrin glue (23). In some cases, human amniotic membrane graft is applied first followed by SLET. The hypothesis supporting SLET is that LSCs in each piece of graft can proliferate and form epithelial clump or island, which can then merge with each other to reconstruct corneal epithelial layer. LSCs may remain around the grafted area or slowly migrate toward the limbus (24). In a long-term (median: 1.5 years, range: 1 to 4 years) outcome analysis of 125 cases of SLET in unilateral ocular surface burn, 76% of eyes maintained successful outcomes and 67% of successful cases achieved 20/60 or better vision (25). In addition, analysis of 10 cases underwent penetrating keratoplasty for further visual gain revealed that basal cells expressing limbal stem cell markers [ΔNp63α(+)/ABCG2(+)] were observed in the trephined cornea after undergoing SLET (25).
In general, autografts have high success rates and better long-term prognosis. However, when LSCs cannot be obtained from the patient because of advanced bilateral LSCD, the use of allogeneic cells may be considered if the patient can tolerate systemic immunosuppression. The major limitation of an allograft is the risk of graft rejection because of the high vascular nature at the limbus. Allogeneic limbal stem cells can be used for conjunctival limbal allograft (CLAL), allo-SLET, allo-CLET, and keratolimbal allograft (KLAL).
If patients cannot tolerate long-term immunosuppressive treatment owing to their general conditions, surgical methods using cells from other body parts of the patient, even those without LSCs, should be considered. Cultivated oral mucosal epithelial transplantation (COMET) can be used to harvest autologous oral mucosal cells, to culture cells in vitro on carriers such as the amniotic membrane, or as a cell sheet for transplanting on the diseased ocular surface (26,27).
Cabral et al. (28) have analyzed clinical outcomes of COMET studies published from 2004 to 2019. Among 243 eyes from 24 studies, 70.8% of eyes achieved a stable ocular surface. Visual acuity improvement was observed in 63.5% of eyes. It is known that the presence of pre-operative epithelial defect and poor tear production can increase the failure rate of COMET. Therefore, these abnormalities should be corrected prior to surgery for better outcome. Although several studies reported that the oral mucosal cells grafted with COMET could transdifferentiate into corneal epithelial phenotype expressing cytokeratin such K12 (29,30) without expressing MUC5AC, it is still difficult to definitively determine whether cultivated oral mucosal epithelial cells have transdifferentiated to the corneal lineage or whether the presence of corneal epithelial cells indicates expansion and migration of the remaining corneal cells.
With advances in stem cell technology, other stem cell populations can be transdifferentiated to mimic corneal epithelial cells. Although this is still at the laboratory level, it is a promising option for future treatment of LSCD.
Figure 1 shows an example of how to select an appropriate surgical method according to various patient conditions. The flow chart was modified from suggestions of two recent publications (3,20).
Alternative cells to replace LSCs
With the advancement of stem cell technology, various attempts to use alternative cell populations to replace allogeneic cells are underway. These alternative cell populations can be utilized as limbal stem cell substitutes for ocular surface reconstruction. Pluripotent stem cells, oral mucosa cells, dental pulp stem cells, and hair follicle cells can be cultured on the carrier substrate and transplanted to the eye with severe LSCD. The implanted cell population can transdifferentiate into more corneal epithelial-like cells and maintain ocular surface integrity and transparency. Although the usefulness of these cells is still being verified in the preclinical stages, corneal limbus reconstruction using these cells is possible in the future (Table 3).
Table 3
| Alternative cells | Description | Stage | References |
|---|---|---|---|
| Oral mucosal cells | Autologous oral mucosal cells are cultured on amniotic membrane or as a cell sheet and transplanted (COMET) | Clinical | (26,27,31) |
| Clinical evidence of efficacy has been accumulated. However, oral mucosal cells are thicker and less transparent than corneal epithelial cells. Transplanted oral mucosal epithelial cells have been reported to survive up to 4 years after surgery | |||
| Human pluripotent stem cells | Human embryonic stem cells differentiate into corneal epithelial progenitor cells in conditions mimicking the LSC niche. Conditioned medium, feeder cells, amniotic membrane, and growth factors have been used to produce and LSC niche-mimicking environment | Under clinical trials | (32-36) |
| Human embryonic stem cell-derived corneal epithelial-like cells and endothelial-like cells were co-cultured on an acellular porcine corneal matrix and transplanted in rabbits. The intact and transparent graft was maintained for 8 weeks | |||
| Corneal epithelial cells were generated from induced pluripotent stem cells derived from human dermal fibroblasts. The cell expressed specific corneal epithelium-related genes such as K12, K3, and Pax6 | |||
| Mesenchymal stem cells | MSCs play immunomodulatory roles. They migrate to the injured area of the cornea and decrease inflammatory cell infiltration and cytokine production in animal models | Under clinical trials | (37-39) |
| MSCs can be delivered to the ocular surface either by MSC containing carriers transplantation or direct subconjunctival injection | |||
| Subconjunctival injection of MSCs was effective in the acute phase of severe ocular burn | |||
| Amniotic epithelial cells | Amniotic epithelial cells can differentiate into corneal epithelial-like cells when they are co-cultured with human corneal epithelial cells | Preclinical | (40,41) |
| Hair follicle cells | A LSC niche-mimicking environment induces a cornea epithelial phenotype in stem cells isolated from the hair follicles of mice | Preclinical | (42) |
| Dental pulp stem cell | Dental pulp stem cells labeled with green fluorescent Qtracker 525 were seeded onto a pretreated contact lens and delivered to an ex vivo debrided human cornea. Dental pulp stem cells transdifferentiated into corneal epithelial progenitors and expressed keratin 12 | Preclinical | (43) |
LSCD, limbal epithelial stem cell deficiency; COMET, cultivated oral mucosal epithelial transplantation; LSC, limbal epithelial stem cell; MSCs, mesenchymal stem cells.
Among alternative cells, mesenchymal stem cells (MSCs) deserve further explanation. In general, MSCs can be obtained from bone marrow or adipose tissues of patients. Transplanted MSCs can differentiate into corneal epithelial cells (44). Although a sufficient number of MSCs do not differentiate into corneal epithelial-like cells, transplanted MSCs are known to have anti-angiogenic and anti-inflammatory properties (45). In addition, the secretomes of cultured limbal MSCs can promote corneal epithelial regeneration (46). MSCs found in the normal limbal stem cell niche are believed to be able to crosstalk with LSCs and control LSC proliferation and differentiation (3,47). Therefore, an exogenous supply of MSCs is expected to secrete trophic and growth factors to stimulate residual LSCs, to control inflammation preventing further damage to LSCs, and finally to improve the limbal niche microenvironment. The non-immunogenic nature of MSCs allows them to be used allogeneically without eliciting immunosuppression of the recipient. Several routes of MSCs application in LSCD are possible. Intravenous injection of MSC in acute phase of cornea damage can alleviate ocular signs such as opacity and neovascularization in animal models (48,49). Various carriers including amniotic membrane, contact lens, and biocompatible polymers can be used to deliver MSCs on the ocular surface (50-52). However, the actual number of transplanted MSCs in this method is known to be small compared to injection methods. Subconjunctival injection of MSCs has also been proven to be effective in LSCD (37). MSCs can migrate to the inflammatory area from the subconjunctival space and modulate inflammation and tissue damage. There is still debate whether injected MSCs can change to LSCs.
Reprogrammed cells called induced pluripotent stem cells (iPSCs) are also promising stem cell sources to replace LSCs (32,53,54). iPSCs can be induced by culture in a cocktail of signaling factors. LSCs induced from iPSCs may allow personalized therapy according to the patient’s need without the risk of allogeneic rejection. Recently, iPSC derived corneal epithelial sheet have been transplanted to a patient suffering bilateral LSCD in Japan (55). Although clinical results have not yet been reported, this clinical trial shows clear advances in LSCD treatment.
Keratoprosthesis for advanced bilateral LSCD
When bilateral LSCD is combined with severe dry eye, fornix adhesion, lid abnormality, or previous failures of LSC transplantation, the success of LSC transplantation is limited and the use of keratoprostheses can be considered (12,56). Keratoprostheses do not require long-term systemic immunosuppression. Early visual rehabilitation is another advantage of keratoprostheses. However, risks of retinal detachment, endophthalmitis, glaucoma, and implant extrusion are serious limitations (56). In keratoprosthesis surgery, epithelialization of the corneal surface by the corneal epithelium is abandoned. Instead, an implant is inserted to replace the optically transparent cornea, allowing light to reach the retina. After keratoprosthesis surgery, the ocular surface is totally covered with the conjunctival epithelium except for a small optical opening in the implant. The most popular keratoprostheses are Boston KPro, AutoKPro, LV Pradad KPro, and modified osteo-odonto-keratoprosthesis (MOOKP). Among these, Boston KPro type 1 is the most commonly used keratoprosthesis. It has an optical cylinder with a skirt of donor tissue. Good outcomes of visual rehabilitation have been reported with Boston KPro type. However, retroprosthetic membrane and corneal melting are common complications (57,58). In a retrospective analysis of 23 eyes receiving Boston type 1 keratoprosthesis, postoperative corrective visual acuity of 20/50 or better was obtained in 67% of eyes at a 3-year follow up and persistent corneal epithelial defect and corneal necrosis were observed in 56.5% and 30% of eyes, respectively (59).
Limitations of this review
This review described basic knowledge of LSC and various treatment options for LSCD. Since the purpose of this review was to convey knowledge that a general ophthalmologist or ophthalmology resident could understand, contents that were too specialized or experimental had to be excluded. In addition, only studies published in English and full-length articles retrieved from PubMed search were included in this review. This approach has a limitation in that it does not include latest research studies presented as abstracts in recent academic conferences.
Conclusions
Our understanding of LSCs and LSCD has deepened over the past years. However, much details of LSCs or LSCs niche remains unknown as interesting future topics of research. Despite the enigmatic identity of LSCs, various treatments have been devised and proven effective in clinical practice. However, it should be kept in mind that the best chance of treating LSCD is in the early stages of the disease. In some cases, initial treatment is difficult, especially for hereditary cases, cases with frequent relapses, and cases with intractable underlying etiologies. Once LSCD has progressed, treatment becomes challenging. Nevertheless, in the early treatment of LSCD, every effort should be made to preserve as many LSCs as possible.
Acknowledgments
Funding: None.
Footnote
Provenance and Peer Review: This article was commissioned by the Guest Editors (Joann Kang and Roy S. Chuck) for the series “Ocular Surface Reconstruction/Transplantation” published in Annals of Eye Science. The article has undergone external peer review.
Reporting Checklist: The author has completed the Narrative Review reporting checklist. Available at https://aes.amegroups.com/article/view/10.21037/aes-22-51/rc
Conflicts of Interest: The author has completed the ICMJE uniform disclosure form (available at https://aes.amegroups.com/article/view/10.21037/aes-22-51/coif). The series “Ocular Surface Reconstruction/Transplantation” was commissioned by the editorial office without any funding or sponsorship. The author has no other conflicts of interest to declare.
Ethical Statement:
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Kenyon KR, Tseng SC. Limbal autograft transplantation for ocular surface disorders. Ophthalmology 1989;96:709-22; discussion 722-3. [Crossref] [PubMed]
- Vazirani J, Nair D, Shanbhag S, et al. Limbal Stem Cell Deficiency-Demography and Underlying Causes. Am J Ophthalmol 2018;188:99-103. [Crossref] [PubMed]
- Elhusseiny AM, Soleimani M, Eleiwa TK, et al. Current and Emerging Therapies for Limbal Stem Cell Deficiency. Stem Cells Transl Med 2022;11:259-68. [Crossref] [PubMed]
- Cotsarelis G, Cheng SZ, Dong G, et al. Existence of slow-cycling limbal epithelial basal cells that can be preferentially stimulated to proliferate: implications on epithelial stem cells. Cell 1989;57:201-9. [Crossref] [PubMed]
- Haagdorens M, Van Acker SI, Van Gerwen V, et al. Limbal Stem Cell Deficiency: Current Treatment Options and Emerging Therapies. Stem Cells Int 2016;2016:9798374. [Crossref] [PubMed]
- Bi YL, Bock F, Zhou Q, et al. Central corneal epithelium self-healing after ring-shaped glycerin-cryopreserved lamellar keratoplasty in Terrien marginal degeneration. Int J Ophthalmol 2013;6:251-2. [PubMed]
- Majo F, Rochat A, Nicolas M, et al. Oligopotent stem cells are distributed throughout the mammalian ocular surface. Nature 2008;456:250-4. [Crossref] [PubMed]
- Dua HS, Miri A, Alomar T, et al. The role of limbal stem cells in corneal epithelial maintenance: testing the dogma. Ophthalmology 2009;116:856-63. [Crossref] [PubMed]
- Joe AW, Yeung SN. Concise review: identifying limbal stem cells: classical concepts and new challenges. Stem Cells Transl Med 2014;3:318-22. [Crossref] [PubMed]
- Ordonez P, Di Girolamo N. Limbal epithelial stem cells: role of the niche microenvironment. Stem Cells 2012;30:100-7. [Crossref] [PubMed]
- Li W, Hayashida Y, Chen YT, et al. Niche regulation of corneal epithelial stem cells at the limbus. Cell Res 2007;17:26-36. [Crossref] [PubMed]
- Kate A, Basu S. A Review of the Diagnosis and Treatment of Limbal Stem Cell Deficiency. Front Med (Lausanne) 2022;9:836009. [Crossref] [PubMed]
- Barbaro V, Ferrari S, Fasolo A, et al. Evaluation of ocular surface disorders: a new diagnostic tool based on impression cytology and confocal laser scanning microscopy. Br J Ophthalmol 2010;94:926-32. [Crossref] [PubMed]
- Poli M, Janin H, Justin V, et al. Keratin 13 immunostaining in corneal impression cytology for the diagnosis of limbal stem cell deficiency. Invest Ophthalmol Vis Sci 2011;52:9411-5. [Crossref] [PubMed]
- Ramirez-Miranda A, Nakatsu MN, Zarei-Ghanavati S, et al. Keratin 13 is a more specific marker of conjunctival epithelium than keratin 19. Mol Vis 2011;17:1652-61. [PubMed]
- Nubile M, Lanzini M, Miri A, et al. In vivo confocal microscopy in diagnosis of limbal stem cell deficiency. Am J Ophthalmol 2013;155:220-32. [Crossref] [PubMed]
- Banayan N, Georgeon C, Grieve K, et al. Spectral-domain Optical Coherence Tomography in Limbal Stem Cell Deficiency. A Case-Control Study. Am J Ophthalmol 2018;190:179-90. [Crossref] [PubMed]
- Lee WD, Devarajan K, Chua J, et al. Optical coherence tomography angiography for the anterior segment. Eye Vis (Lond) 2019;6:4. [Crossref] [PubMed]
- Liu L, Hartwig D, Harloff S, et al. Corneal epitheliotrophic capacity of three different blood-derived preparations. Invest Ophthalmol Vis Sci 2006;47:2438-44. [Crossref] [PubMed]
- Fernandez-Buenaga R, Aiello F, Zaher SS, et al. Twenty years of limbal epithelial therapy: an update on managing limbal stem cell deficiency. BMJ Open Ophthalmol 2018;3:e000164. [Crossref] [PubMed]
- Holland EJ. Management of Limbal Stem Cell Deficiency: A Historical Perspective, Past, Present, and Future. Cornea 2015;34:S9-15. [Crossref] [PubMed]
- Sangwan VS, Sharp JAH. Simple limbal epithelial transplantation. Curr Opin Ophthalmol 2017;28:382-6. [Crossref] [PubMed]
- Sangwan VS, Basu S, MacNeil S, et al. Simple limbal epithelial transplantation (SLET): a novel surgical technique for the treatment of unilateral limbal stem cell deficiency. Br J Ophthalmol 2012;96:931-4. [Crossref] [PubMed]
- Shanbhag SS, Patel CN, Goyal R, et al. Simple limbal epithelial transplantation (SLET): Review of indications, surgical technique, mechanism, outcomes, limitations, and impact. Indian J Ophthalmol 2019;67:1265-77. [Crossref] [PubMed]
- Basu S, Sureka SP, Shanbhag SS, et al. Simple Limbal Epithelial Transplantation: Long-Term Clinical Outcomes in 125 Cases of Unilateral Chronic Ocular Surface Burns. Ophthalmology 2016;123:1000-10. [Crossref] [PubMed]
- Nakamura T, Inatomi T, Sotozono C, et al. Transplantation of cultivated autologous oral mucosal epithelial cells in patients with severe ocular surface disorders. Br J Ophthalmol 2004;88:1280-4. [Crossref] [PubMed]
- Nishida K, Yamato M, Hayashida Y, et al. Corneal reconstruction with tissue-engineered cell sheets composed of autologous oral mucosal epithelium. N Engl J Med 2004;351:1187-96. [Crossref] [PubMed]
- Cabral JV, Jackson CJ, Utheim TP, et al. Ex vivo cultivated oral mucosal epithelial cell transplantation for limbal stem cell deficiency: a review. Stem Cell Res Ther 2020;11:301. [Crossref] [PubMed]
- Kim YJ, Lee HJ, Ryu JS, et al. Prospective Clinical Trial of Corneal Reconstruction With Biomaterial-Free Cultured Oral Mucosal Epithelial Cell Sheets. Cornea 2018;37:76-83. [Crossref] [PubMed]
- Kolli S, Ahmad S, Mudhar HS, et al. Successful application of ex vivo expanded human autologous oral mucosal epithelium for the treatment of total bilateral limbal stem cell deficiency. Stem Cells 2014;32:2135-46. [Crossref] [PubMed]
- Ma DH, Hsueh YJ, Ma KS, et al. Long-term survival of cultivated oral mucosal epithelial cells in human cornea: generating cell sheets using an animal product-free culture protocol. Stem Cell Res Ther 2021;12:524. [Crossref] [PubMed]
- Hayashi R, Ishikawa Y, Ito M, et al. Generation of corneal epithelial cells from induced pluripotent stem cells derived from human dermal fibroblast and corneal limbal epithelium. PLoS One 2012;7:e45435. [Crossref] [PubMed]
- Zhang C, Du L, Pang K, et al. Differentiation of human embryonic stem cells into corneal epithelial progenitor cells under defined conditions. PLoS One 2017;12:e0183303. [Crossref] [PubMed]
- Ahmad S, Stewart R, Yung S, et al. Differentiation of human embryonic stem cells into corneal epithelial-like cells by in vitro replication of the corneal epithelial stem cell niche. Stem Cells 2007;25:1145-55. [Crossref] [PubMed]
- Zhang C, Du L, Sun P, et al. Construction of tissue-engineered full-thickness cornea substitute using limbal epithelial cell-like and corneal endothelial cell-like cells derived from human embryonic stem cells. Biomaterials 2017;124:180-94. [Crossref] [PubMed]
- Vattulainen M, Ilmarinen T, Viheriälä T, et al. Corneal epithelial differentiation of human pluripotent stem cells generates ABCB5+ and ∆Np63α+ cells with limbal cell characteristics and high wound healing capacity. Stem Cell Res Ther 2021;12:609. [Crossref] [PubMed]
- Galindo S, de la Mata A, López-Paniagua M, et al. Subconjunctival injection of mesenchymal stem cells for corneal failure due to limbal stem cell deficiency: state of the art. Stem Cell Res Ther 2021;12:60. [Crossref] [PubMed]
- Liang L, Luo X, Zhang J, et al. Safety and feasibility of subconjunctival injection of mesenchymal stem cells for acute severe ocular burns: A single-arm study. Ocul Surf 2021;22:103-9. [Crossref] [PubMed]
- Shukla S, Shanbhag SS, Tavakkoli F, et al. Limbal Epithelial and Mesenchymal Stem Cell Therapy for Corneal Regeneration. Curr Eye Res 2020;45:265-77. [Crossref] [PubMed]
- Yao M, Chen J, Yang XX, et al. Differentiation of human amniotic epithelial cells into corneal epithelial-like cells in vitro. Int J Ophthalmol 2013;6:564-72. [PubMed]
- Zhou Q, Liu XY, Ruan YX, et al. Construction of corneal epithelium with human amniotic epithelial cells and repair of limbal deficiency in rabbit models. Hum Cell 2015;28:22-36. [Crossref] [PubMed]
- Meyer-Blazejewska EA, Call MK, Yamanaka O, et al. From hair to cornea: toward the therapeutic use of hair follicle-derived stem cells in the treatment of limbal stem cell deficiency. Stem Cells 2011;29:57-66. [Crossref] [PubMed]
- Kushnerev E, Shawcross SG, Sothirachagan S, et al. Regeneration of Corneal Epithelium With Dental Pulp Stem Cells Using a Contact Lens Delivery System. Invest Ophthalmol Vis Sci 2016;57:5192-9. [Crossref] [PubMed]
- Rohaina CM, Then KY, Ng AM, et al. Reconstruction of limbal stem cell deficient corneal surface with induced human bone marrow mesenchymal stem cells on amniotic membrane. Transl Res 2014;163:200-10. [Crossref] [PubMed]
- Eslani M, Putra I, Shen X, et al. Corneal Mesenchymal Stromal Cells Are Directly Antiangiogenic via PEDF and sFLT-1. Invest Ophthalmol Vis Sci 2017;58:5507-17. [Crossref] [PubMed]
- Shibata S, Hayashi R, Okubo T, et al. The secretome of adipose-derived mesenchymal stem cells attenuates epithelial-mesenchymal transition in human corneal epithelium. Regen Ther 2019;11:114-22. [Crossref] [PubMed]
- Yazdanpanah G, Haq Z, Kang K, et al. Strategies for reconstructing the limbal stem cell niche. Ocul Surf 2019;17:230-40. [Crossref] [PubMed]
- Mittal SK, Omoto M, Amouzegar A, et al. Restoration of Corneal Transparency by Mesenchymal Stem Cells. Stem Cell Reports 2016;7:583-90. [Crossref] [PubMed]
- Ye J, Yao K, Kim JC. Mesenchymal stem cell transplantation in a rabbit corneal alkali burn model: engraftment and involvement in wound healing. Eye (Lond) 2006;20:482-90. [Crossref] [PubMed]
- Calonge M, Pérez I, Galindo S, et al. A proof-of-concept clinical trial using mesenchymal stem cells for the treatment of corneal epithelial stem cell deficiency. Transl Res 2019;206:18-40. [Crossref] [PubMed]
- Holan V, Trosan P, Cejka C, et al. A Comparative Study of the Therapeutic Potential of Mesenchymal Stem Cells and Limbal Epithelial Stem Cells for Ocular Surface Reconstruction. Stem Cells Transl Med 2015;4:1052-63. [Crossref] [PubMed]
- Espandar L, Caldwell D, Watson R, et al. Application of adipose-derived stem cells on scleral contact lens carrier in an animal model of severe acute alkaline burn. Eye Contact Lens 2014;40:243-7. [Crossref] [PubMed]
- Takahashi K, Yamanaka S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell 2006;126:663-76. [Crossref] [PubMed]
- Yu D, Chen M, Sun X, et al. Differentiation of mouse induced pluripotent stem cells into corneal epithelial-like cells. Cell Biol Int 2013;37:87-94. [Crossref] [PubMed]
- Watanabe S, Hayashi R, Sasamoto Y, et al. Human iPS cells engender corneal epithelial stem cells with holoclone-forming capabilities. iScience 2021;24:102688. [Crossref] [PubMed]
- Vazirani J, Mariappan I, Ramamurthy S, et al. Surgical Management of Bilateral Limbal Stem Cell Deficiency. Ocul Surf 2016;14:350-64. [Crossref] [PubMed]
- Wang LQ, Wu TY, Chen XN, et al. Long-term outcomes of Boston keratoprosthesis type I: the Chinese People's Liberation Army General Hospital experience. Br J Ophthalmol 2022;106:781-5. [Crossref] [PubMed]
- Samarawickrama C, Strouthidis N, Wilkins MR. Boston keratoprosthesis type 1: outcomes of the first 38 cases performed at Moorfields Eye Hospital. Eye (Lond) 2018;32:1087-92. [Crossref] [PubMed]
- Sejpal K, Yu F, Aldave AJ. The Boston keratoprosthesis in the management of corneal limbal stem cell deficiency. Cornea 2011;30:1187-94. [Crossref] [PubMed]
Cite this article as: Park CY. An updated narrative review of treatment for limbal epithelial stem cell deficiency. Ann Eye Sci 2023;8:15.


