Association of sensorineural hearing loss and pseudoexfoliation syndrome: a meta-analysis
Introduction
Pseudoexfoliation syndrome (PEX), also known as exfoliation syndrome, is an age-related disease characterized by the abnormal production and deposition of pseudo-exfoliation fibrous material, which mainly occurs in the front of the eye (1). Diffuse pseudo-exfoliation deposits are believed to be the cause of many ocular and extraocular complications, such as glaucoma, lens opacity, cataract surgery complications, retinal vascular occlusion, and age-related macular degeneration (2-5). PEX is now considered a systemic disease because these substances are also present in other parts of the body, including the skin, vascular structures and internal organs (such as the heart, kidneys, gallbladder, and lungs) and inner ears (6).
The inner ear is a complex organ. Like the structure of the anterior segment of the eye, the tectorial membrane and basement membrane of the inner ear come from the neuroectoderm. According to previous studies, these membranes in the inner ear may also be where the pseudo-exfoliation material deposits (7). The accumulation of this substance on these structures will affect the function of the inner ear and cause hearing loss.
Many epidemiological and experimental studies have evaluated the relationship between sensorineural hearing loss (SNHL) and PEX, but the results are inconsistent. Some studies (8-19) have shown an association between PEX and increased risk of SNHL, while other studies (20,21) have shown no association. In view of the possibility of diagnosing PEX through slit lamp examination to identify individuals at increased risk of SNHL, research on this issue may have important clinical significance. In addition, it is of special importance for the early diagnosis and treatment of patients with hearing and vision loss (double sensory loss). In view of the evidence from recent studies, the goal of this study is to assess the correlation between PEX and SNHL risk through a systematic review and meta-analysis of all available epidemiological studies. This meta-analysis adopts a standardized program and is conducted in accordance with the PRISMA reporting checklist (available at https://aes.amegroups.com/article/view/10.21037/aes-21-68/rc) (22).
Methods
Literature search
We searched PubMed, Embase, and ISI Web of Science through computers and identified relevant publications. The retrieval time was from 1966 to November 2020. Both the title and open text fields are used to identify the article, with no application date or language restrictions. Search for the following medical terms and their combinations: (I) PEX, exfoliative syndrome; (II) hearing impairment, hearing dysfunction, hearing disorder, hearing loss, ear symptoms. The related article function is also used, and the reference list of all retrieved studies and related reviews is supplemented by manual search to expand the search scope. If multiple articles are published using the same case series, only the most complete series of studies will be included.
Inclusion and exclusion criteria
Related research needs to meet the following criteria: (I) a case-control study to assess the correlation between SNHL and PEX; (II) an unrelated case-control design; (III) an odds ratio (OR) with a 95% confidence interval (CI) or other data that can be used to estimate OR (95% CI). SNHL is defined as any low, medium, and high frequency hearing impairment. Exclusion criteria include: unpublished papers, non-human studies, letters/case reports, editorials, comments, studies lacking searchable raw data, studies lacking a suitable control group, and studies using insufficient case definitions.
Data extraction
Research data was extracted by two independent researchers using a standardized data collection form, and if there are differences, they will be discussed. The information collected from each study includes: first author and year of publication, country of origin, subject ethnicity, study design, exposure and evaluation results, number of cases and control groups, correlation, point estimate and 95% CI, and any adjustment/stratification/matching variables.
Quality assessment
The quality of the included studies was independently assessed by the same two investigators using the Newcastle-Ottawa Scale (NOS) (23). NOS uses a “star” rating system to judge the quality of research based on three aspects: selection, comparability and exposure (case-control study) or results (cohort study). The score ranges from 0 (worst) to 9 (best). The higher the score, the better the methodological quality. A score of ≥7 is considered high quality.
Statistical analysis
In this meta-analysis, the correlation between SNHL and PEX was estimated by calculating combined ORs and 95% CI. A random effects model was used to conservatively estimate the effects of potential population differences in the study using the methods of Der Simonian and Laird. The significance of the combined OR was determined by the Z test (P<0.05 is considered statistically significant).
The heterogeneity is determined by Q test and I2 statistics. I2 values of more than 50% are defined as having obvious heterogeneity. For the Q statistic, a P value <0.1 is considered statistically heterogeneity. Through subgroup analysis based on race, case composition, sample method, sample size, age and gender matching, and hearing loss definitions, the size of combined OR in each class and their respective heterogeneity tests were examined. Through meta-regression analysis, the influence of these variables on the heterogeneity of cross-stratigraphic research is studied. The reliability of the results of the meta-analysis was determined by omitting one study for each round of sensitivity analysis. The Begg funnel chart and Egger test were used to statistically evaluate publication bias. The possible publication bias is represented by an asymmetric graph. A P value of less than 0.05 in the Egger test is considered to represent a statistically significant publication bias (24). All analyses were performed using STATA software version 11.2 (Stata, Texas College Station).
Results
Literature search and characteristics
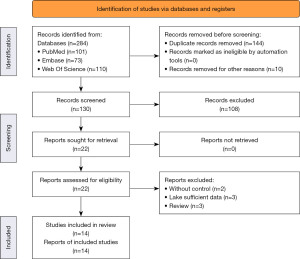
The detailed process steps of selecting included documents are shown in Figure 1. The initial search found 284 potentially relevant studies. Through the electronic database to delete duplicate content and delete irrelevant topics, leaving 22 full-text articles for further qualification evaluation. Eight articles were excluded for the following reasons: review articles (n=3) (25-27); insufficient article data (n=3) (28-30); lack of control group (n=2) (31,32). In the end, 14 articles met the inclusion criteria and were included in this meta-analysis (8-21).
Table 1 lists the design characteristics and participant characteristics of the included studies. All studies are case-control designs. Of the 14 articles identified, 2 were population-based and 12 were based on hospital research. The research areas are: six studies were conducted in Turkey, three studies were conducted in Iran, and one each was conducted in Poland, Iceland, Malaysia, Greece and Egypt. These studies involved 1,142 PEX patients and 9,914 control patients. The average score of the NOS results was 7.57 (range, 7 to 9), indicating that the quality of each research methodology is good.
Table 1
| Study (first author) | Year of publication | Country | Study design | No. of case | No. of control | Definition of sensorineural hearing loss | Covariates adjusted | NOS score |
|---|---|---|---|---|---|---|---|---|
| Yildirim | 2017 | Turkey | PB | 100 | 1,909 | NA | Sex | 7 |
| Temporale | 2016 | Poland | HB | 28 | 23 | With hearing thresholds >25 dB hearing level | Age, sex | 8 |
| Tryggvason | 2016 | Iceland | PB | 158 | 123 | defined by the Liden-Jerger procedure | Age, education, lifestyle, chronic diseases | 8 |
| Singham | 2014 | Malaysia | HB | 68 | 55 | With hearing thresholds >20 dB hearing level | Age, sex | 8 |
| Papadopoulos | 2012 | Greece | HB | 47 | 22 | Defined by the American National Standards Institute (ANSI) 1969 | Age, sex | 8 |
| Samarai | 2012 | Iran | HB | 50 | 50 | Defined by the ISO7029 standard | Age, sex | 8 |
| Zojaji | 2011 | Iran | HB | 33 | 33 | With hearing thresholds >20 dB hearing level | Age, sex | 8 |
| Shazly | 2011 | Egypt | HB | 320 | 7,418 | NA | Age, sex | 7 |
| Turgut | 2010 | Turkey | HB | 34 | 40 | With hearing thresholds >20 dB hearing level | Age | 7 |
| Erbek | 2009 | Turkey | HB | 32 | 23 | NA | Age, sex | 7 |
| Yazdani | 2008 | Iran | HB | 83 | 83 | defined by the ISO7029 standard | Age, sex | 8 |
| Ozturk | 2008 | Turkey | HB | 63 | 38 | With hearing thresholds >25 dB hearing level | Age | 7 |
| Turacli | 2007 | Turkey | HB | 51 | 22 | With hearing thresholds >20 dB hearing level | Age | 7 |
| Aydoğan Ozkan | 2006 | Turkey | HB | 75 | 75 | With hearing thresholds >25 dB hearing level | Age, sex, systemic diseases | 9 |
PB, population based; HB, hospital based; NOS, Newcastle-Ottawa Scale; NA, data not available.
Association between SNHL and PEX
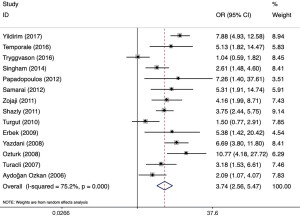
The forest diagram (Figure 2) shows the correlation between SNHL and PEX. Overall, compared with the control group, the risk of hearing loss in PEX patients was significantly increased [OR: 3.74 (95% CI: 2.56 to 5.47); P<0.001]. However, the included studies have obvious heterogeneity (P<0.001, I2=75.2%).
Subgroup, sensitivity analyses, and meta-regression
We further explored the heterogeneity between SNHL and PEX studies through subgroup analysis and sensitivity analysis. A hierarchical analysis of subgroups based on race, case composition, sample method, sample size, age and gender matching, and hearing loss definitions revealed that there is a significant relationship between SNHL and PEX in all subgroups. Subgroup analysis also showed that heterogeneity was reduced in studies that matched age and gender and used the same definition of hearing loss. In addition, in the sensitivity analysis, one study was omitted at a time, and the combined OR of the remaining studies was calculated, and consistent results were obtained. The combined ORs were statistically significant and the results were similar. The OR ranged from 3.45 (95% CI: 2.38 to 5.01) to 4.16 (95% CI: 3.01 to 5.76). Meta-regression analysis explored the impact of key characteristics of the study (i.e., subgroup factors) on heterogeneity, and the results showed that the definition of hearing loss was the key to the heterogeneity (P=0.044) (Table 2).
Table 2
| Groups | Studies (n) | OR | 95% CI | P (overall effect) | Heterogeneity | P for meta-regression | ||
|---|---|---|---|---|---|---|---|---|
| Lower bound | Upper bound | I2 (%) | P | |||||
| Overall | 14 | 3.74 | 2.56 | 5.47 | <0.001 | 75.2 | <0.001 | – |
| Ethnicity | 0.570 | |||||||
| Caucasian | 13 | 3.88 | 2.57 | 5.86 | <0.001 | 76.4 | <0.001 | |
| Asian | 1 | 2.61 | 1.48 | 4.60 | 0.001 | – | – | |
| Case component | 0.189 | |||||||
| PEX | 6 | 5.22 | 2.57 | 10.58 | <0.001 | 73.6 | 0.002 | |
| PEX/PEXG | 8 | 3.09 | 2.02 | 4.73 | <0.001 | 73.0 | 0.001 | |
| Sample method | 0.474 | |||||||
| PB | 2 | 2.88 | 0.39 | 20.97 | 0.297 | 96.6 | <0.001 | |
| HB | 12 | 3.82 | 2.79 | 5.23 | <0.001 | 51.2 | 0.020 | |
| Sample size | 0.840 | |||||||
| <100 | 6 | 3.35 | 2.10 | 5.36 | <0.001 | 35.9 | 0.168 | |
| ≥100 | 8 | 3.87 | 2.28 | 6.59 | <0.001 | 84.0 | <0.001 | |
| Age and sex matching | 0.544 | |||||||
| Yes | 9 | 3.92 | 2.97 | 5.18 | <0.001 | 21.9 | 0.249 | |
| No | 5 | 3.28 | 1.31 | 8.23 | 0.011 | 90.3 | <0.001 | |
| Definition of hearing loss | 0.044 | |||||||
| >20 | 4 | 2.62 | 1.74 | 3.95 | <0.001 | 33.1 | 0.214 | |
| >25 | 3 | 4.65 | 1.69 | 12.84 | 0.003 | 6.8 | 0.300 | |
| IOS7029 | 2 | 6.34 | 3.86 | 10.41 | <0.001 | 0 | 0.698 | |
| Others | 5 | 3.83 | 1.65 | 8.88 | 0.002 | 87.0 | <0.001 | |
PEX, pseudoexfoliation syndrome; PEXG, pseudoexfoliation glaucoma; PB, population based; HB, hospital based; OR, odds ratio; CI, confidence interval.
Publication bias
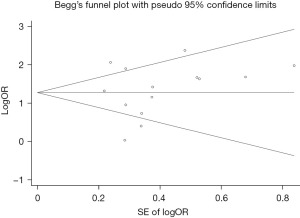
This meta-analysis uses the Begg funnel chart and the Egger test to assess publication bias. Begg’s funnel chart did not show any obvious evidence of asymmetry, and Egger’s test did not detect publication bias (P=0.663). These results indicate that the publication bias in the SNHL and PEX studies is not statistically significant in this meta-analysis (Figure 3).
Discussion
Sensory disturbances are common in the elderly. Hearing and vision loss usually increase with age. More and more scientific evidence shows that the frequency of simultaneous loss of hearing and vision is more frequently than would be expected (33). Therefore, further research on the relationship between hearing and vision loss is very important, which may have important public health and clinical significance. To the best of our knowledge, this study is the first meta-analysis to investigate the relationship between PEX and hearing loss. Our results show that PEX increases the incidence of SNHL by 2.74 times.
SHNL is caused by damage to cochlear cells and/or surrounding structures, as well as disturbances in the quality of auditory nerve stimulation and/or the central nervous system's understanding of the quality of stimulation. The underlying mechanism between PEX and SNHL is not fully understood, but several possible theories and hypotheses have been proposed. First, both the inner ear basement membrane and the anterior eye membrane are formed by the ectoderm. Some studies have found that both the inner ear cover membrane and the basement membrane have pseudo-exfoliation substances (7). Pseudo-exfoliated substances deposited on the cochlear basement membrane and/or covering membrane may cause changes in the chemical composition of the surrounding environment, resulting in uneven energy transmission to the nerve sensory hair cells (31). Secondly, the deposition of pseudo-exfoliation material on the blood vessel wall may damage the blood supply of the inner ear and temporal lobe, leading to dysfunction of the auditory receptors and auditory cortex (18). Third, the pathology and biochemistry of these diseases may be similar, or they may have common susceptibility factors, such as age, cellular oxidative stress, and insufficient cytoprotective mechanisms (11,19,34). Fourth, previous studies demonstrated that PEX patients have an increased risk for abdominal and aortic aneurisms as well as pelvic organ prolapse (35,36). PEX syndrome is very much a disease of the extracellular matrix leading to tissue laxity, which contributes to those 3 disorders. This tissue laxity almost certainly arises from the altered expression and/or activity of LOXL1, an enzyme that is strongly associated with PEX and that cross-links elastic fibers. Altered LOXL1 activity could bring about dysfunction of spiral ligament fibrocytes or structural changes to the extracellular matrix that could significantly impact hearing function (37).
In this study, the results of subgroup analysis and sensitivity analysis were similar and robust, and neither subgroup analysis nor single study significantly changed the correlation. A significant positive correlation was observed in all subgroups except for the subgroup study designed by population base. There is obvious heterogeneity in PEX and SNHL risk studies. Considering the differences in population characteristics, the determination of SNHL, and the adjustment of confounding factors, this is not surprising. Fortunately, in the subgroup analysis, heterogeneity was reduced in studies that matched age and gender and used the same definition of hearing loss. Both PEX and SNHL are age-related diseases. SNHL is very common and is closely related to male gender and age (20). In some analytical studies, due to the lack of adjustment for age and gender, their results may be biased. In addition, our meta-regression analysis shows that the definition of hearing loss also leads to heterogeneity to a large extent, which indicates that different definitions of hearing loss and the determination of hearing loss may have an important impact on the results of these studies.
Meta-analysis has advantages over individual research, but our research has some potential limitations. First, since only a few studies meet our eligibility criteria, we have assembled the results of studies using different designs and different definitions of hearing loss. Second, most studies are conducted in the Middle East, so these results may not be generalized to other regions. Third, the included study is a case-control design, so the causal relationship between PEX and SNHL cannot be determined.
Despite the limitations of this study, this meta-analysis presents some valuable findings. It provides aggregated data on a large number of cases and controls in order to better understand the association between PEX and SNHL risks. In addition, this research extends several questions. The first question is whether PEX has a causal effect on SNHL, or whether it is just a surrogate marker for other biological risk factors. This problem should be solved by considering several issues, including the interval between the onset of the two diseases, the use of standardized hearing loss definitions, and control measures for confounding factors, etc. Second, what is the exact mechanism by which PEX increases the risk of hearing loss? Further research, including well-designed longitudinal clinical trials and basic scientific research, is necessary to address these issues in order to better understand this connection and provide convincing evidence for clinical practice to prevent hearing disorders.
Conclusions
This meta-analysis shows an association between PEX and increased risk of SNHL. The effect of PEX on the prevalence and severity of hearing loss indicates that PEX is clearly a systemic disease that includes otological complications, in addition to the multiple other disorders that have been demonstrated to occur at higher frequency in individuals with PEX syndrome.
Acknowledgments
Funding: The study was supported by the National Natural Science Foundation of China (No. 82000890), Hainan Provincial Natural Science Foundation of China (No. 820RC780), Young Talents’ Science and Technology Innovation Project of Hainan Association for Science and Technology (No. QCXM202020), and Hainan Province Clinical Medical Center.
Footnote
Reporting Checklist: The authors have completed the PRISMA reporting checklist. Available at https://aes.amegroups.com/article/view/10.21037/aes-21-68/rc
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://aes.amegroups.com/article/view/10.21037/aes-21-68/coif). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Schlötzer-Schrehardt U, Khor CC. Pseudoexfoliation syndrome and glaucoma: from genes to disease mechanisms. Curr Opin Ophthalmol 2021;32:118-28. [Crossref] [PubMed]
- Ritch R. Exfoliation syndrome-the most common identifiable cause of open-angle glaucoma. J Glaucoma 1994;3:176-7. [Crossref] [PubMed]
- Puska P, Tarkkanen A. Exfoliation syndrome as a risk factor for cataract development: five-year follow-up of lens opacities in exfoliation syndrome. J Cataract Refract Surg 2001;27:1992-8. [Crossref] [PubMed]
- Gillies WE, Brooks AM. Central retinal vein occlusion in pseudoexfoliation of the lens capsule. Clin Exp Ophthalmol 2002;30:176-87. [Crossref] [PubMed]
- Kling F, Colin J. Potential association of pseudoexfoliation syndrome (PEX) with age-related macular degeneration (ARMD). J Fr Ophtalmol 2001;24:7-12. [PubMed]
- Schlötzer-Schrehardt U, Naumann GO. Ocular and systemic pseudoexfoliation syndrome. Am J Ophthalmol 2006;141:921-37. [Crossref] [PubMed]
- Lim DJ. Functional structure of the organ of Corti: a review. Hear Res 1986;22:117-46. [Crossref] [PubMed]
- Yildirim N, Yasar E, Gursoy H, et al. Prevalence of pseudoexfoliation syndrome and its association with ocular and systemic diseases in Eskisehir, Turkey. Int J Ophthalmol 2017;10:128-34. [PubMed]
- Temporale H, Karasińska-Kłodowska A, Turno-Kręcicka A, et al. Evaluating the Hearing of Patients with Pseudoexfoliation Syndrome. Adv Clin Exp Med 2016;25:1215-21. [Crossref] [PubMed]
- Singham NV, Zahari M, Peyman M, et al. Association between Ocular Pseudoexfoliation and Sensorineural Hearing Loss. J Ophthalmol 2014;2014:825936. [Crossref] [PubMed]
- Papadopoulos TA, Charalabopoulou M, Vathylakis I, et al. Prevalence and severity of sensorineural hearing loss in patients with exfoliation syndrome. Eur Rev Med Pharmacol Sci 2012;16:902-7. [PubMed]
- Samarai V, Samarei R, Haghighi N, et al. Sensory-neural hearing loss in pseudoexfoliation syndrome. Int J Ophthalmol 2012;5:393-6. [PubMed]
- Zojaji R, Alesheykh A, Sedaghat MR, et al. Pseudoexfoliation syndrome and sensorineural hearing loss. Iran J Otorhinolaryngol 2011;23:149-58. [PubMed]
- Shazly TA, Farrag AN, Kamel A, et al. Prevalence of pseudoexfoliation syndrome and pseudoexfoliation glaucoma in Upper Egypt. BMC Ophthalmol 2011;11:18. [Crossref] [PubMed]
- Erbek S, Erbek SS, Karalezli A, et al. Function of outer hair cells in patients with pseudoexfoliation. Kulak Burun Bogaz Ihtis Derg 2009;19:130-3. [PubMed]
- Yazdani S, Tousi A, Pakravan M, et al. Sensorineural hearing loss in pseudoexfoliation syndrome. Ophthalmology 2008;115:425-9. [Crossref] [PubMed]
- Ozturk F, Kurt E, Inan UU, et al. Is pseudoexfoliation associated with sensorineural hearing loss? Neurosciences (Riyadh) 2008;13:61-4. [PubMed]
- Turacli ME, Ozdemir FA, Tekeli O, et al. Sensorineural hearing loss in pseudoexfoliation. Can J Ophthalmol 2007;42:56-9. [Crossref] [PubMed]
- Aydoğan Ozkan B, Yüksel N, Keskin G, et al. Homocysteine levels in plasma and sensorineural hearing loss in patients with pseudoexfoliation syndrome. Eur J Ophthalmol 2006;16:542-7. [Crossref] [PubMed]
- Tryggvason G, Jonasson F, Cotch MF, et al. Hearing in older adults with exfoliation syndrome/exfoliation glaucoma or primary open-angle glaucoma. Acta Ophthalmol 2016;94:140-6. [Crossref] [PubMed]
- Turgut B, Alpay HC, Kaya MK, et al. The evaluation of vestibular functions in patients with pseudoexfoliation syndrome. Eur Arch Otorhinolaryngol 2010;267:523-7. [Crossref] [PubMed]
- Moher D, Liberati A, Tetzlaff J, et al. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med 2009;6:e1000097. [Crossref] [PubMed]
- Stang A. Critical evaluation of the Newcastle-Ottawa scale for the assessment of the quality of nonrandomized studies in meta-analyses. Eur J Epidemiol 2010;25:603-5. [Crossref] [PubMed]
- Egger M, Davey Smith G, Schneider M, et al. Bias in meta-analysis detected by a simple, graphical test. BMJ 1997;315:629-34. [Crossref] [PubMed]
- Ritch R. Ocular and systemic manifestations of exfoliation syndrome. J Glaucoma 2014;23:S1-8. [Crossref] [PubMed]
- Bettis DI, Allingham RR, Wirostko BM. Systemic diseases associated with exfoliation syndrome. Int Ophthalmol Clin 2014;54:15-28. [Crossref] [PubMed]
- Ritch R. Systemic Associations of Exfoliation Syndrome. Asia Pac J Ophthalmol (Phila) 2016;5:45-50. [Crossref] [PubMed]
- Detorakis ET, Chrysochoou F, Paliobei V, et al. Evaluation of the acoustic function in pseudoexfoliation syndrome and exfoliation glaucoma: audiometric and tympanometric findings. Eur J Ophthalmol 2008;18:71-6. [Crossref] [PubMed]
- Paliobei VP, Psillas GK, Mikropoulos DG, et al. Hearing Evaluation in Patients with Exfoliative and Primary Open-Angle Glaucoma. Otolaryngol Head Neck Surg 2011;145:125-30. [Crossref] [PubMed]
- Papadopoulos TA, Naxakis SS, Charalabopoulou M, et al. Exfoliation syndrome related to sensorineural hearing loss. Clin Exp Ophthalmol 2010;38:456-61. [Crossref] [PubMed]
- Cahill M, Early A, Stack S, et al. Pseudoexfoliation and sensorineural hearing loss. Eye (Lond) 2002;16:261-6. [Crossref] [PubMed]
- Shaban RI, Asfour WM. Ocular pseudoexfoliation associated with hearing loss. Saudi Med J 2004;25:1254-7. [PubMed]
- Kremmer S, Anastassiou G, Selbach JM. Hearing Disorders with Glaucoma. Laryngorhinootologie 2016;95:755-61. [PubMed]
- Seidman MD, Ahmad N, Bai U. Molecular mechanisms of age-related hearing loss. Ageing Res Rev 2002;1:331-43. [Crossref] [PubMed]
- Pompoco CJ, Curtin K, Taylor S, et al. Summary of Utah Project on Exfoliation Syndrome (UPEXS): using a large database to identify systemic comorbidities. BMJ Open Ophthalmol 2021;6:e000803. [Crossref] [PubMed]
- Wirostko B, Allingham R, Wong J, et al. Utah Project on Exfoliation Syndrome (UPEXS): Insight Into Systemic Diseases Associated With Exfoliation Syndrome. J Glaucoma 2018;27:S75-7. [Crossref] [PubMed]
- Peeleman N, Verdoodt D, Ponsaerts P, et al. On the Role of Fibrocytes and the Extracellular Matrix in the Physiology and Pathophysiology of the Spiral Ligament. Front Neurol 2020;11:580639. [Crossref] [PubMed]
Cite this article as: Huang W, Kuang J, Chen F, Fu Y. Association of sensorineural hearing loss and pseudoexfoliation syndrome: a meta-analysis. Ann Eye Sci 2022;7:26.