
Experimental models of retinopathy of prematurity
Introduction
Retinopathy of prematurity (ROP) is the most common cause of blindness affecting preterm infants (1). Similarly to other proliferative retinopathies, ROP is characterized by the disruption of normal angiogenic mechanisms leading to retinal neovascularization (NV) (1). In premature infants, the interrupted development of the retinal vasculature at the time of birth, together with an initial hyperoxia-induced microvascular obliteration, determine the onset of a peripheral avascular area (2). These vascular changes result in an increased hypoxia in the avascular areas, due to the gap between oxygen supply and increased metabolic demands of the developing retina (2). As a result, the massive upregulation of pro-angiogenic factors promotes the abnormal retinal NV, characterized by the formation of leaky, non-organized vessels, called pre-retinal tufts, which grow into the vitreous cavity and lead to visual loss following retinal detachment (2). Pre-retinal tufts can regress in case of adequate oxygen administration (2). However, although about 90% of ROP infants suffer from only mild visual impairment, patients with untreated NV are still at an elevated risk for progression to more severe conditions, promoting impaired vision and blindness (1). Several multifactorial agents characterize the pathogenesis of ROP, involving the crosstalk among different pathophysiological factors, such as hypoxia, inflammation and oxidative pathways (3).
Animal models of oxygen-induced retinopathy (OIR) are popular models widely used to reproduce ROP and investigate pathologic NV in the retina, as well as the involved molecular mechanisms and new putative therapies. Several common OIR models were established in different species, such as feline (4), murine (5,6), and canine (7), all of which mimicking ROP in humans. A fundamental prerequisite for an animal OIR is an immature retinal vasculature at birth with an active physiological angiogenesis, similar to the human retina before 31 weeks of gestation (1). The exposure to constant or cycling hyperoxia in OIR models leads to the arrest/retardation of normal retinal vascular development, vascular dropout and following retinal NV, thus reliably reproducing the two phases of ROP (1). Although the reproducibility of NV is different across species, the easy induction of NV, together with the rapid visualization and quantification of pre-retinal tufts, makes OIR models useful tools for the study of mechanisms and potential therapeutics against ischemic retinopathies, including ROP (8). To date, the most commonly used OIR models are performed in rodents, such as mice and rats, which are discussed in the following sections. Smith’s mice OIR (6) and Penn’s rat OIR model (5) protocols are described. Following the description of the protocols, a brief overview of several outcomes deriving from the application of the procedures described below is given. In this respect, practical suggestions about techniques which could be used for each aspect are provided, as well as a brief description of exemplificative results deriving from the application of such methods.
Protocols
Required material
- A balance to weight the animals.
- An isolated chamber connected to a tube system.
- An oxygen sensor (Pro-Custom Elettronica, Milano, Italy).
- An oxygen pressure monitor (Teledyne system, Analytical Instruments, City of Industry, CA, USA).
- An oxygen tank (Siad, Bergamo, Italy).
- A nitrogen tank (only for rat protocol) (Siad).
- A soda lime with a color indicator system or a filtration system.
- A thermometer.
- A humidity monitors.
- Sodium pentobarbital (30 mg/kg).
A schematic representation of an example of how a custom-made chamber could be set up is shown in Figure 1.
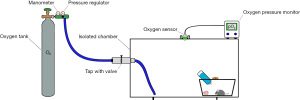
Mouse OIR model
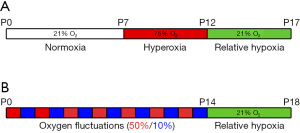
A schematic representation of the protocol is shown in Figure 2A.
- Set up a hyperoxia chamber according to the manufacturer’s instructions or build a custom-made chamber provided with an oxygen sensor and a tube system where oxygen can be inserted into the cage. Calibrate the O2 sensor with 100% O2 and normal room air according to the manufacturer’s instructions.
- Record the date of birth of the animals [indicated as postnatal day 0 (P0)]. Leave the pups with their nursing mother in room air in a separate cage for 7 days.
- At P7, record the weight of the developing pups before placing them in the OIR chamber.
- Note 1: it is advisable to record the weight of the OIR exposed pups during the key points of the protocol: before and after hyperoxia; at the time of sacrifice. Indeed, it has been demonstrated that the extent of the NV is highly influenced by the postnatal weight gain of the pups. Pups weighting <5 g at P17 have delayed and markedly prolonged vaso-obliteration and NV compared to those with medium (5–7.5 g) and extensive (>7.5 g) weight gain (9). Thus, it is recommended not to use pups under 6 g at P17 due to the possible altered vascular phenotype (10). Moreover, animals from at least 3 litters should be considered to overcome the variability within single litters (10).
- Place the P7 mice into OIR chamber and provide oxygen until a partial pressure of 75% is reached. Keep the mice in the OIR chamber until P12. Check the oxygen pressure regularly and, if the O2 levels decrease, add more oxygen to restore the pressure at 75%. Add soda lime with a color indicator to the bottom of the chamber to avoid the excessive CO2 accumulation. Alternatively, use a filtration system. Monitor the humidity and temperature inside the chamber and maintain the humidity at 40–65%. If required, place dishes filled with water on the bottom of the chamber to increase the humidity. Check the wellbeing of the animals and always provide enough food and water.
- Note 2: it is advisable to keep extra lactating dams available to possibly replace the nursing dams in case of exhaustion. Keep the litter size to 6–7 pups for each dam if C57BL/6J mice is used, since pups in litters larger than that tend to have restricted weight gain (10).
- Note 3: nursing mothers could suffer during hyperoxia exposure. This could result in poor nutrition of the pups and cannibalism episodes (10). Therefore, it is advisable to regularly check the state of the litter and decide the number of pups per litter considering these side effects. The substitution with a surrogate in case of poor maternal care/nursing mother’s death should be considered. To reduce the detrimental effects of hyperoxia on nursing mothers, the hyperoxia paradigm could be modified to 22 hours of high oxygen and 2–3 hours of room air exposure per day, rather than continuous high oxygen exposure (11). Although these changes lead the same to OIR phenotypes, it is recommended to consider the possibility that hypoxic episodes during the hyperoxia phase might influence the extent of the avascular area and the degree of NV (12).
- At P12 remove the mouse cages from the chamber and weight the animals.
- Leave the animals in room air until P17 and then proceed with the sacrifice through lethal injection of sodium pentobarbital. Mice should be weighed before proceeding with the sacrifice.
- Note 4: possible treatment paradigms during the OIR protocol could be possible to perform. Drug administration in OIR commonly occurs by either intravitreal or intraperitoneal injection. The treatment paradigm should be chosen depending on the experimental setup and the focus of the study. Indeed, the time frame of the treatment could change depending on the necessity to study the effects on vaso-obliteration (P7–P12) or NV (P12–P17). For intravitreal injection, inject the drug preferably in only one eye, keeping the contralateral eye as a control. Choose no more than 1.0 µL as injection volume and consider that mice open their eyes at P14. For intraperitoneal injections, choose the correct dose and constantly measure the weight of the animals to decide the injection volume, given that animals are still under development (consider no more than 100 µL for the injection).
Rat OIR model
A schematic representation of the protocol is shown in Figure 2B. The protocol described here is the result of several changes applied over time to have a model mimicking more closely the clinical settings of ROP. From an initial exposure of the rat pups at continuous 80% O2, the hyperoxia exposure paradigm was changed to 80%/40% cycles and, finally, to alternating 50%/10% before keeping the animals to room air (13). Moreover, also the timing of the oxygen exposures has been progressively changed over the different protocols (13). The 50/10 rat model described here has been considered to more closely reflect the arterial blood gases experienced by a sick premature infant and result in a greater retardation of retinal blood vessel development, as well as in an increased NV severity compared to other rat models (13).
- Set up the chamber following the manufacturer’s instructions or build a custom-made chamber connected to an oxygen sensor and a tube system where oxygen or nitrogen can be added into the cage. Calibrate the O2 sensor with 100% oxygen and/or room air.
- Record the date of birth of the animals (indicated as P0). Pups and their nursing mother should be placed in the OIR chamber within 4 hours from birth.
- Before placing the animals in the OIR chamber, weight the pups.
- Note 5: it is advisable to record the weight of the OIR exposed pups during the key points of the protocol: before and after oxygen fluctuations; before the sacrifice. Similar to the mouse model, postnatal growth and the degree of NV are directly correlated in the rat OIR (13).
- Place newborn P0 rats into OIR chamber and provide oxygen until a partial pressure of 50% is reached. Monitor the pressure through the oxygen sensor connected to the oxygen pressure monitor. Check the pressure 2/3 times daily and adjust the pressure if oxygen levels decrease during the day. Add soda lime with a color indicator in the chamber or use a filtration system to absorb excessive CO2. Check the humidity and the temperature in the chamber and constantly keep the humidity between 40% and 65%. If an increased humidity is needed, place dishes with water at the bottom of the chamber.
- Note 6: for rat OIR protocols, the use of high litter size, a minimum of 14 pups/dam, is recommended to have sufficient NV induction, since in the rat model NV occurs at a lower grade (8,14). Indeed, large litters are advantageous because they limit the amount of nutrition each pup receives, adversely affect their body weight, and therefore increase severity of NV (13). To obtain a huge litter size, pool pups from several time-matched litters. To account for potential effects within a single litter, rats from a minimum of 2/3 separate litters should be considered for the analyses (15,16).
- After 24 hours reduce oxygen pressure to 10% by adding nitrogen. Monitor the pressure through the oxygen sensor connected to the oxygen pressure monitor. Check the pressure 2/3 times daily and adjust the pressure by adding O2 if oxygen levels decrease during the day.
- Note 7: if directly eliminating the humidity in the chamber is not possible, regularly open the chamber once daily to absorb the condensed water on the chamber walls. Given that fluctuations between 50% and 10% should be performed, this strategy could be used when oxygen pressures need to be changed in order to spare oxygen and nitrogen. Keep the animals in 21% atmosphere for a short time, adequately to remove the humidity. Place then the pups in the chamber and newly regulate oxygen pressure to the corresponding pressure based on daily fluctuations.
- Repeat the alternation of 50% and 10% oxygen every 24 hours until P14. Check the oxygen pressure regularly and, if the O2 levels decrease, add more oxygen to restore the pressure at 50% or 10% depending on the phase. Check the wellbeing of the animals and always provide enough food and water for the entire duration of oxygen fluctuations.
- Note 8: to our experience, nursing rat mothers are more resistant to OIR conditions than mice. Therefore, cannibalism phenomena are rare, but we recommend a careful check of litter wellbeing and consider the idea of a surrogate in case the nursing mother dies in OIR chamber.
- At P14 remove the rat cages from the chamber and weight the animals.
- Leave the animals in room air until P18/P20 and then proceed with the sacrifice through a lethal injection of sodium pentobarbital. Rats should be weighed before proceeding with the sacrifice.
- Note 9: the efficacy of treatment strategies could be tested during the rat OIR protocol. Commonly used drug administration route is managed through either intravitreal or intraperitoneal injection. Treatment protocol depends on the experimental design and the aim of the study. Indeed, the administration times could change depending on whether the experimenter wants to evaluate the effect on peripheral avascular area (P0–P14) or NV (P14–P18/P20). For intravitreal injections, it is advisable to inject the drug in only one eye, maintaining the contralateral eye as a control. Inject up to 1.0 µL of solution and consider that rats open their eyes at P14. For intraperitoneal injections, use the correct dose according to animal weight, which should be constantly measured before the injection, since these animals are still under development (consider up to 100 µL to inject).
Examples of outcomes deriving from rodent OIR models
Retinal dysfunction
ROP is known to result in long-term effects on vision, which have been observed in patients even after the active phases of the disease (17). Functional alterations of the retina following an OIR protocol could be assessed by electroretinogram (ERG). In particular, a full-field ERG could be used to evaluate the activity of the entire retina in animals after the exposure to OIR protocols (18). This non-invasive analysis can be conducted in vivo under physiological or nearly physiological conditions through electrodes collocated on the corneal surface. The positive and negative components of the recorded ERG wave indicate the sum of the potentials originating from different stages of retinal processing after a provided light stimulus (18). ERG analyses should be performed under systemic anesthesia, and the further application of local anesthetics is recommended to avoid blinking. ERG in developing pups should be recorded at later stages than P14, given that the animals open their eyes by the end of the second post-natal week. Thus, it is recommended to perform functional analysis at least P17/P18, when an adequate eye opening has occurred favoring the correct electrodes positioning and the wide opening of the vision field. To evaluate retinal function, a dark-adapted scotopic ERG could be recorded (18). A scotopic ERG wave usually comprises of two components, the initial negative-going a-wave (mainly from photoreceptors) and the following positive-going b-wave, generated by post-receptor cells (18). Evidence of impaired ERG responses has been reported both in OIR mice and rats. In particular, functional impairment can be observed during the hypoxia phase, evidenced by the reduced a- and b-wave amplitudes in OIR animals (19,20). Moreover, functional deficits in OIR models persist even after vascular regression (21,22).
Retinal avascular areas and pathological NV
The visualization of retinal vasculature on flat mounts represents the gold standard method to evaluate any sign of vessel alteration induced by the OIR protocol. Retinal samples could be fixed and labelled with specific molecules to recognize endothelial markers, thus ultimately providing a tool to label retinal vessels (Figure 3). The most commonly used vascular labelling is performed with isolectin B4 (IB4), which allows vascular visualization by targeting the surface of endothelial cells (14). The whole flat mount analysis allows the experimenter to have a full contemporary view of the retinal vascularization, thus providing an ideal instrument to quantify OIR vascular outcomes (11). Indeed, avascular areas and pre-retinal tufts could be quantified through image-processing software (i.e., Photoshop, Image J) starting from a retinal flat mount image acquired through the microscope. For example, vaso-obliterated areas could be measured by outlining the avascular area related to total retinal area (23,24). On the other hand, pre-retinal tufts could be measured either manually or semi automatedly and be expressed as a percentage of total retinal area (23). Alternatively, intravitreous NV in the rat OIR model could be analyzed by counting the number of clock hours involved or summing areas of NV (24).
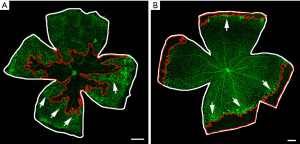
In an OIR mouse model, a central avascular area can be observed at P12, as a consequence of the hyperoxia-mediated vaso-obliteration (11). As a response to ischemic conditions after returning to room air, the significant NV leads to the growth of neovascular tufts from P14 to onward, reaching the peak intensity at P17 (11). Thus, the visualization and quantification of NV on retinal flat mounts is mostly performed at P17. After this time point, abnormal NV begins a spontaneous gradual regression, completely disappearing by approximately P25, when almost neither vaso-obliterated areas nor NV are visible (23).
Unlike the mouse model, in the rat OIR retinas the exposure to cycling levels of oxygen slows the retinal vascular development in peripheric areas. Indeed, by P14 the 50/10 OIR retinas display a peripheral avascular area, compared to the fully vascularized retinas of room air pups (24). This time point, which correspond to the end of oxygen fluctuations, could be used to analyze the formation of the peripheral avascular area, a typical hallmark of the protocol success. In the rat OIR model, NV occurring at the boundary between vascularized and avascular areas in the midperipheral retina reaches its peak at P18/P20 (24). Similar to the mouse model, NV recovers by P25, thus to observe differences in retinal vascularization it is recommended to end the experiment at P20 (14).
Expression of pro-angiogenic factors
Vascular dysfunctions characterizing OIR retinas occur as a consequence of the dysregulated expression of pro-angiogenic factors due to hypoxia conditions (2). Molecular analyses represent a useful tool to assess the changes in the levels of pro-angiogenic factors occurring at either transcriptional or translational level during the OIR protocol. It should be considered that the small dimension of neonatal pup retinas may hinder the molecular experiments, due to the unavailability of sufficient tissue to provide acceptable material for the analysis. Thus, samples containing pooled retinas from more than one animal are often required to obtain adequate protein or RNA levels for quantification (11).
In a mouse OIR model, a downregulation of pro-angiogenic factors, such as vascular endothelial growth factor (VEGF) and erythropoietin (Epo), is observed at P12 after hyperoxia (1,23). In contrast, during NV an increase in expression of pro-angiogenic factors, such as VEGF, Epo, insulin growth factor 1 (IGF-1), fibroblast growth factor 2 (FGF2), heparin-binding EGF-like growth factor (HB-EGF), and hepatocyte growth factor (HGF), as well as the activation of VEGF, Epo and IGF-1 receptor pathways, are reported as a consequence of hypoxia (23,25-29). In contrast, in the rat OIR model an upregulation of VEGF and VEGFR-2 levels has been reported both at the end of oxygen fluctuations (P14) and retinal NV (P18) (24,30).
Retinal inflammation and glial activation
The activation of inflammatory mechanisms is another pathological feature of OIR models. Indeed, increasing evidence shows that a local and/or systemic inflammation might contribute as a risk factor in ROP, and the increment of inflammatory molecules has been demonstrated to perturb retinal vessel development and induce pathological features of ROP (31). The increased expression of pro-inflammatory molecules could be assessed through molecular analyses from retinal extracts, both at transcriptional and protein levels. In this respect, in an OIR mouse model several pro-inflammatory molecules have been observed to increase during the early stages of hypoxia, with a following decline during the vascular regression phase (32).
Furthermore, inflammation is often accompanied by Müller cell gliosis and microglial/macrophages activation, increasing the wide spectrum of mechanisms damaging OIR retinas (33). Indeed, these cellular types have been demonstrated to be involved in the production of inflammatory molecules and the mediation of retinal damage (34). An immunohistochemical analysis on retinal sections could be used as an example to visualize these cell types in the retina during its inflamed status. In this respect, markers labeling glial cells could be used to visualize the localization of the cells and their activation state. For example, the frequently used marker to label gliotic Müller cells is glial fibrillary acidic protein (GFAP), whereas ionized calcium-binding adapter molecule 1 (Iba1) is the most commonly used marker to detect retinal microglial cells and recruited macrophages. The upregulation of GFAP, as well as the increase in GFAP positive processes among the entire retina, are documented in an OIR model during hypoxia, suggesting the activation of Müller cells (35). Moreover, an increase in GFAP immunoreactive processes underlining the activation of Müller cells is documented also in the OIR rat model (36). Consistently, an increase in ameboid shaped Iba1 positive cells, resembling the morphology of activated microglia/macrophages, has been described in mice OIR retinas (35). In this respect, when performing microglial/macrophagic labeling, it is recommended to add the use of other markers which could indicate the subpopulation and the activation state of microglia/macrophages, since different phenotypes of these cells might help either retinal NV or revascularization and disease resolution (32).
Retinal morphological alterations
Vascular alterations and retinal inflammation could lead to retinal cell damage, ultimately resulting in retinal cell death. The overall increase in retinal cell death during an OIR protocol could be indicated by the reduction in the thickness of retinal layers, assessed through histological analysis after the labeling of cell nuclei by 4',6-diamidino-2-phenylindole (DAPI) counterstaining or through histological stains to visualize retinal layers. In addition, to assess morphological alterations on photoreceptor outer segments, histological analyses using specific markers labeling such portions could be useful to evaluate putative shortening or disorganizations of these segments (35). A decrease in total retinal thickness is observed in both mouse and rat OIR models, especially in the inner retinal layers which seems to be the most affected by the changes in oxygen pressures (19,37). Nonetheless, some evidence reported also damage to the outer retina, with reduced outer nuclear layer (ONL) thickness and decreased photoreceptor density, especially in the rat model from later stages (P60) (38,39). Moreover, a shorter and disorganized structure of photoreceptor outer segments has been reported in both models, which might partially explain the functional impairment of the ERG a-wave (35,40). Furthermore, evidence of apoptotic microvascular cells, such as endothelial cells and pericytes, has been documented (41). The alterations in retinal thickness persist also during late stages after oxygen exposure (21).
Choroidal degeneration
Emerging evidence has underlined that the choroid is also extensively affected during the development of ROP both in animal models and in humans (33). The effects derived from the application of OIR protocol on choroidal vasculature could be evaluated by histological analyses to assess choroidal thickness. An example of such analysis could be performed by the staining of retinal/choroidal sections through markers labeling the endothelial cells of choroidal vessels (38,39). Alternatively, molecular analyses of cell death markers, at either transcriptional or protein levels, could be performed from choroidal tissues extracts to assess some basic molecular events associated with choroidal involution (38).
In the rat OIR model, a choroidal thinning was observed at P14 following oxygen fluctuations, which persisted during retinal NV phase and over time up to P210 (38,39). Concomitantly, the upregulation of cell death markers has been observed during oxygen fluctuations and NV phases (38).
Discussion and conclusions
ROP is one of the most common reasons of blindness occurring during prenatal stages (3). Therefore, an appropriate model able to reproduce faithfully the main features of the disease, such as vascular dysfunction, inflammation, retinal damage, choroidal involution and functional impairment, might represent a useful tool to study pathophysiological mechanisms and possible therapeutical strategies. In this context, the OIR models provide a bona fide representation of the most relevant events associated with ROP, thus offering a valid instrument to analyze the mechanisms involved in the disease and some hypothetical resolutions. Actually, most of the information about molecular mechanisms of the disease, as well as the effects of several drugs, come from these models, underlining the importance of these animals in ROP research.
The most important prerequisite of an animal OIR is represented by an immature retinal vasculature at birth, whose postnatal development could be easily modulated to mimic vascular abnormalities typical of human ROP (1). Unlike humans, where the retinal vasculature matures before a baby is born term, these animals develop their retinal vascular system during the first postnatal stages, thus resembling the condition of a human preterm retina (1). In this respect, the exposure to either constant hyperoxia or oxygen fluctuations during these early phases of development in different species reproduce an initial vaso-obliteration, followed by abnormal NV during the return in normoxic atmosphere, reliably reproducing the phases of human disease (1). Since the neovascular response is very reproducible and quantifiable, the study of the effects of antiproliferative and antiangiogenic treatments could be easily quantified in these models. Considering that OIR models are not only characterized by vascular alterations typical of the human disease, but are also affected by inflammation, retinal cell death, choroidal degeneration and functional loss, these models might provide all the elements which could be considered as hallmarks to evaluate the effect of a putative protective treatment. Indeed, these emerging pathophysiological events have been also observed in ROP patients, where local and systemic inflammation, choroidal thinning and long-term functional impairment might occur in the pathogenesis of the disease (17,33). Therefore, OIR models might represent a helpful tool to study these mechanisms and the efficacy of novel promising therapeutical strategies applied to these factors more deeply.
The OIR mouse model by Smith (6) and the rat model by Penn (5) are the most extensively used for studying the mechanisms involved in pathological angiogenesis, as well as the effects of various angiostatics or neuroprotectants (8). However, although both models could reproduce basic characteristics of the disease, there are some differences in phenotypic outcomes, especially in vascular defects. Indeed, while in the mouse OIR model the vaso-obliteration during the exposure to hyperoxia involves central vessels, the oxygen fluctuations lead to the formation of an avascular area in retinal periphery (8). Therefore, in this aspect, the rat OIR model reproduces more faithfully what happens in human ROP, providing a more reliable model to use when evaluating preventive effects aiming at promoting the revascularization of non-perfused periphery. Nevertheless, the mouse OIR model reliably reproduces the pathologic events occurring during ischemia-induced NV (8). The levels of observed NV are higher compared with the rat model, supporting its use in the screening of antiangiogenic compounds (8). Moreover, another advantage of using mice is the relatively higher ease of genetic manipulation to study directly the role of different genes in NV. For instance, mice OIR models carrying a deletion of beta-adrenergic receptors 1 and 2, were useful to study the role of adrenergic system in pathological angiogenesis and find a promising therapeutical target of disease (42).
Despite the wide usage of these models in pre-clinical research of ROP, there are several limitations connected to important factors which could influence the variability of OIR outcomes. Indeed, strain-dependent differences in terms of NV severity have been reported both in mice and rats, where the extension of avascular areas and NV differs among strains (11,43). Moreover, variability is further promoted if animals belonging to the same strain come from different vendors (11,13). In addition, another source of variability can be found among different mouse litters, since different numerosity and nursing conditions could lead to diverse health of pups, postnatal weight gain and then OIR phenotype severity (11,13). Therefore, all these variables should be addressed in order to obtain a higher reproducibility in obtaining OIR phenotypes. As another relevant point, other complications of premature birth are not reproduced by OIR models since protocols are conducted postnatally with healthy animals without all the comorbidities typically of human preterm infants (3,11). In this respect, healthy animals display a mature lung physiology, which might derive in systemic (and presumably retinal) oxygen tensions that might not be similar to the clinical setting. The OIR rat model has addressed this issue by using fluctuating oxygen exposures in ranges that produce more clinically representative arterial oxygen pressures. However, the timing is much different and less random from that connected with the variable respiratory function in preterm infants with immature lungs (44). Finally, another limitation of the most commonly used OIR models in rodents is represented by the fact that these animals are not characterized by some features of ROP, such as retinal detachment (8).
Despite OIR models are characterized by several factors which could influence the variability in their outcomes, the appliance of OIR models in ROP research has played a pivotal role so far, and with the new advances in genetic manipulation, a deeper understanding of the role of new genes in ROP pathogenesis could be possible. Moreover, the versatility of these models due to the reproduction of a reliable human-like NV phenotype, makes the OIR models useful for the study of other ischemic retinopathies such as diabetic retinopathy (DR). Indeed, the current rodent models of DR (either genetic or drug-induced) are able to reproduce only the early non-proliferative stages, whereas they fail to develop the NV associated to the proliferative phase, likely due to the short life span of the animals (8). Since the mechanisms behind NV and tuft formation are similar between ROP and DR, OIR models could represent a valid alternative to study the dynamics characterizing the proliferative stages of DR and the potential of antiangiogenic drugs appliable to both the diseases. Therefore, the potential of these models to be a compensatory model to study the angiogenic response in other proliferative retinopathies, makes OIR models even more valuable in research of retinal diseases.
In conclusion, OIR models display several characteristics also confirmed in human patients, which make these animals a bona fide tool in the pre-clinical research of ROP and other proliferative retinopathies. Nonetheless, there are many limitations and many variables which should be considered whilst using these models to obtain proper results. In this respect, this study protocol tries to provide starting guidelines critically introducing the basic knowledge of OIR protocols, the related outcomes and its variables and limitations. This study protocol has the aim to provide a critical tool to colleagues who want to start with the OIR studying, and a helpful instrument for a critical beginning of their work, which will be further deepened by the literature regarding the specific topic they will choose.
Acknowledgments
I really thank Dr. Rosario Amato for the critical reading of this manuscript and the fundamental suggestions.
Funding: None.
Footnote
Provenance and Peer Review: This article was commissioned by the Guest Editor (Dario Rusciano) for the series “Preclinical Models in Ophthalmic Research” published in Annals of Eye Science. The article has undergone external peer review.
Conflicts of Interest: The author has completed the ICMJE uniform disclosure form (available at https://aes.amegroups.com/article/view/10.21037/aes-21-49/coif). The series “Preclinical Models in Ophthalmic Research” was commissioned by the editorial office without any funding or sponsorship. The author has no other conflicts of interest to declare.
Ethical Statement: The author is accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Dai C, Webster KA, Bhatt A, et al. Concurrent Physiological and Pathological Angiogenesis in Retinopathy of Prematurity and Emerging Therapies. Int J Mol Sci 2021;22:4809. [Crossref] [PubMed]
- Cavallaro G, Filippi L, Bagnoli P, et al. The pathophysiology of retinopathy of prematurity: an update of previous and recent knowledge. Acta Ophthalmol 2014;92:2-20. [Crossref] [PubMed]
- Hartnett ME. Pathophysiology and mechanisms of severe retinopathy of prematurity. Ophthalmology 2015;122:200-10. [Crossref] [PubMed]
- Ashton N, Ward B, Serpell G. Effect of oxygen on developing retinal vessels with particular reference to the problem of retrolental fibroplasia. Br J Ophthalmol 1954;38:397-432. [Crossref] [PubMed]
- Penn JS, Henry MM, Tolman BL. Exposure to alternating hypoxia and hyperoxia causes severe proliferative retinopathy in the newborn rat. Pediatr Res 1994;36:724-31. [Crossref] [PubMed]
- Smith LE, Wesolowski E, McLellan A, et al. Oxygen-induced retinopathy in the mouse. Invest Ophthalmol Vis Sci 1994;35:101-11. [PubMed]
- McLeod DS, Brownstein R, Lutty GA. Vaso-obliteration in the canine model of oxygen-induced retinopathy. Invest Ophthalmol Vis Sci 1996;37:300-11. [PubMed]
- Liu CH, Wang Z, Sun Y, et al. Animal models of ocular angiogenesis: from development to pathologies. FASEB J 2017;31:4665-81. [Crossref] [PubMed]
- Stahl A, Chen J, Sapieha P, et al. Postnatal weight gain modifies severity and functional outcome of oxygen-induced proliferative retinopathy. Am J Pathol 2010;177:2715-23. [Crossref] [PubMed]
- Connor KM, Krah NM, Dennison RJ, et al. Quantification of oxygen-induced retinopathy in the mouse: a model of vessel loss, vessel regrowth and pathological angiogenesis. Nat Protoc 2009;4:1565-73. [Crossref] [PubMed]
- Kim CB, D'Amore PA, Connor KM. Revisiting the mouse model of oxygen-induced retinopathy. Eye Brain 2016;8:67-79. [Crossref] [PubMed]
- Reynaud X, Dorey CK. Extraretinal neovascularization induced by hypoxic episodes in the neonatal rat. Invest Ophthalmol Vis Sci 1994;35:3169-77. [PubMed]
- Barnett JM, Yanni SE, Penn JS. The development of the rat model of retinopathy of prematurity. Doc Ophthalmol 2010;120:3-12. [Crossref] [PubMed]
- Gammons MV, Bates DO. Models of Oxygen Induced Retinopathy in Rodents. Methods Mol Biol 2016;1430:317-32. [Crossref] [PubMed]
- Wang H, Yang Z, Jiang Y, et al. Quantitative analyses of retinal vascular area and density after different methods to reduce VEGF in a rat model of retinopathy of prematurity. Invest Ophthalmol Vis Sci 2014;55:737-44. [Crossref] [PubMed]
- Jiang Y, Wang H, Culp D, et al. Targeting Müller cell-derived VEGF164 to reduce intravitreal neovascularization in the rat model of retinopathy of prematurity. Invest Ophthalmol Vis Sci 2014;55:824-31. [Crossref] [PubMed]
- Hansen RM, Moskowitz A, Akula JD, et al. The neural retina in retinopathy of prematurity. Prog Retin Eye Res 2017;56:32-57. [Crossref] [PubMed]
- Frishman L, Wang MH. Electroretinogram of Human, Monkey and Mouse. In: Levin L. editor. Adler’s physiology of the eye. Philadelphia, PA, USA: Saunders, 2011:480-501.
- Cammalleri M, Locri F, Catalani E, et al. The Beta Adrenergic Receptor Blocker Propranolol Counteracts Retinal Dysfunction in a Mouse Model of Oxygen Induced Retinopathy: Restoring the Balance between Apoptosis and Autophagy. Front Cell Neurosci 2017;11:395. [Crossref] [PubMed]
- Liu K, Akula JD, Falk C, et al. The retinal vasculature and function of the neural retina in a rat model of retinopathy of prematurity. Invest Ophthalmol Vis Sci 2006;47:2639-47. [Crossref] [PubMed]
- Nakamura S, Imai S, Ogishima H, et al. Morphological and functional changes in the retina after chronic oxygen-induced retinopathy. PLoS One 2012;7:e32167. [Crossref] [PubMed]
- Zhang N, Favazza TL, Baglieri AM, et al. The rat with oxygen-induced retinopathy is myopic with low retinal dopamine. Invest Ophthalmol Vis Sci 2013;54:8275-84. [Crossref] [PubMed]
- Stahl A, Connor KM, Sapieha P, et al. The mouse retina as an angiogenesis model. Invest Ophthalmol Vis Sci 2010;51:2813-26. [Crossref] [PubMed]
- Hartnett ME. The effects of oxygen stresses on the development of features of severe retinopathy of prematurity: knowledge from the 50/10 OIR model. Doc Ophthalmol 2010;120:25-39. [Crossref] [PubMed]
- Martini D, Monte MD, Ristori C, et al. Antiangiogenic effects of β2 -adrenergic receptor blockade in a mouse model of oxygen-induced retinopathy. J Neurochem 2011;119:1317-29. [Crossref] [PubMed]
- Ristori C, Filippi L, Dal Monte M, et al. Role of the adrenergic system in a mouse model of oxygen-induced retinopathy: antiangiogenic effects of beta-adrenoreceptor blockade. Invest Ophthalmol Vis Sci 2011;52:155-70. [Crossref] [PubMed]
- Fang L, Barber AJ, Shenberger JS. Regulation of fibroblast growth factor 2 expression in oxygen-induced retinopathy. Invest Ophthalmol Vis Sci 2014;56:207-15. [Crossref] [PubMed]
- Inoue Y, Shimazawa M, Nakamura S, et al. Both Autocrine Signaling and Paracrine Signaling of HB-EGF Enhance Ocular Neovascularization. Arterioscler Thromb Vasc Biol 2018;38:174-85. [Crossref] [PubMed]
- Lorenc VE, Lima E, Silva R, Hackett SF, et al. Hepatocyte growth factor is upregulated in ischemic retina and contributes to retinal vascular leakage and neovascularization. FASEB Bioadv 2020;2:219-33. [Crossref] [PubMed]
- Budd SJ, Thompson H, Hartnett ME. Association of retinal vascular endothelial growth factor with avascular retina in a rat model of retinopathy of prematurity. Arch Ophthalmol 2010;128:1014-21. [Crossref] [PubMed]
- Rivera JC, Dabouz R, Noueihed B, et al. Ischemic Retinopathies: Oxidative Stress and Inflammation. Oxid Med Cell Longev 2017;2017:3940241. [Crossref] [PubMed]
- Li J, Yu S, Lu X, et al. The phase changes of M1/M2 phenotype of microglia/macrophage following oxygen-induced retinopathy in mice. Inflamm Res 2021;70:183-92. [Crossref] [PubMed]
- Rivera JC, Holm M, Austeng D, et al. Retinopathy of prematurity: inflammation, choroidal degeneration, and novel promising therapeutic strategies. J Neuroinflammation 2017;14:165. [Crossref] [PubMed]
- Cuenca N, Fernández-Sánchez L, Campello L, et al. Cellular responses following retinal injuries and therapeutic approaches for neurodegenerative diseases. Prog Retin Eye Res 2014;43:17-75. [Crossref] [PubMed]
- Vessey KA, Wilkinson-Berka JL, Fletcher EL. Characterization of retinal function and glial cell response in a mouse model of oxygen-induced retinopathy. J Comp Neurol 2011;519:506-27. [Crossref] [PubMed]
- Downie LE, Pianta MJ, Vingrys AJ, et al. Neuronal and glial cell changes are determined by retinal vascularization in retinopathy of prematurity. J Comp Neurol 2007;504:404-17. [Crossref] [PubMed]
- Soetikno BT, Yi J, Shah R, et al. Inner retinal oxygen metabolism in the 50/10 oxygen-induced retinopathy model. Sci Rep 2015;5:16752. [Crossref] [PubMed]
- Shao Z, Dorfman AL, Seshadri S, et al. Choroidal involution is a key component of oxygen-induced retinopathy. Invest Ophthalmol Vis Sci 2011;52:6238-48. [Crossref] [PubMed]
- Zhou TE, Rivera JC, Bhosle VK, et al. Choroidal Involution Is Associated with a Progressive Degeneration of the Outer Retinal Function in a Model of Retinopathy of Prematurity: Early Role for IL-1β. Am J Pathol 2016;186:3100-16. [Crossref] [PubMed]
- Fulton AB, Reynaud X, Hansen RM, et al. Rod photoreceptors in infant rats with a history of oxygen exposure. Invest Ophthalmol Vis Sci 1999;40:168-74. [PubMed]
- Saito Y, Geisen P, Uppal A, et al. Inhibition of NAD(P)H oxidase reduces apoptosis and avascular retina in an animal model of retinopathy of prematurity. Mol Vis 2007;13:840-53. [PubMed]
- Dal Monte M, Cammalleri M, Mattei E, et al. Protective effects of β1/2 adrenergic receptor deletion in a model of oxygen-induced retinopathy. Invest Ophthalmol Vis Sci 2014;56:59-73. [Crossref] [PubMed]
- van Wijngaarden P, Coster DJ, Brereton HM, et al. Strain-dependent differences in oxygen-induced retinopathy in the inbred rat. Invest Ophthalmol Vis Sci 2005;46:1445-52. [Crossref] [PubMed]
- Madan A, Penn JS. Animal models of oxygen-induced retinopathy. Front Biosci 2003;8:d1030-43. [Crossref] [PubMed]
Cite this article as: Canovai A. Experimental models of retinopathy of prematurity. Ann Eye Sci 2022;7:18.


