
Update on biologic therapies for juvenile idiopathic arthritis-associated uveitis
Introduction
Juvenile idiopathic arthritis (JIA), previously known as juvenile rheumatoid arthritis (JRA), is the most common rheumatic disease of childhood. The onset of JIA is before age 16, and arthritis is present for a minimum of 6 weeks (1). The most common extra-articular manifestation of JIA is uveitis (JIA-U), which involves inflammation of the uvea comprising the iris, ciliary body, and choroid, and this occurs in up to 20% of children with JIA. JIA-U is the most common cause of non-infectious uveitis in childhood. More broadly, non-infectious uveitis is one of the leading causes of preventable blindness in the United States while also accounting for 15% of the cases of blindness in Western countries (2).
Nearly 21% of all cases of childhood uveitis in the United States have an associated diagnosis of JIA-U while the prevalence of JIA-U in bilateral pediatric uveitis cases in the United States is 89% (3). However, there is significant variation in the prevalence worldwide (4,5). Ocular complications and vision loss have been noted in 3–66% of children with JIA-U with up to 25% of children with JIA-U progressing to blindness (6). Given the severity of the disease and its association with an increased prevalence of blindness in Western countries, regular ophthalmic screening of children with JIA, stratification of risk by JIA subtype, and timely, effective treatment are needed in order to prevent the sequelae of JIA-U that may lead to visual impairment.
This review focuses on standard treatments as well as promising biologic therapies of JIA-U. Although synthetic treatments are commonly administered and have demonstrated efficacy, adverse effects and treatment-refractory disease may require biologic alternatives. With the emergence of biologic therapies with targeted mechanisms, a broad understanding of their efficacy as JIA-U treatment and safety profiles are needed to provide the full complement of therapies for multidisciplinary care from rheumatologists and ophthalmologists.
Clinical presentation
JIA-U can be classified based on the Standardization of Uveitis Nomenclature (SUN) guidelines by specific anatomic location of inflammation: anterior, intermediate, and posterior. Panuveitis involves inflammation of all three of these locations (7). In addition, uveitis can be stratified based on time course into acute, subacute, chronic, or recurrent. The most common form of JIA-U is chronic anterior uveitis with JIA-U accounting for 75% of all pediatric anterior uveitis cases (8). General ocular symptoms may include redness, pain, photophobia, excessive tearing, and floaters (9). Anterior uveitis can present with corneal findings (keratic precipitates) and pupillary changes (e.g., posterior synechiae) with dense white cataract in some individuals (10). Chronic anterior uveitis, however, often presents asymptomatically, which may delay diagnosis and subsequent treatment, thereby increasing the risk of ocular complications.
Screening
Since an asymptomatic presentation can occur and children may have difficulty reporting ocular changes, regular screenings are vital for detection of JIA-U. Various ophthalmic screening guidelines exist, and most include a slit lamp examination, which allows for examination of the anterior segment and posterior segment including the retina and choroid. The American College of Rheumatology/Arthritis Foundation (ACR/AF) guidelines for ophthalmic screening of children with JIA-U recommend ophthalmic screenings every 3 months in patients with JIA with a high risk of developing associated uveitis (11). This frequency is based on the underlying risk factors for JIA-U. The American Academy of Pediatrics (AAP) lists these risk factors for JIA-U as ANA seropositivity, oligoarticular or polyarticular JIA subtypes, early age of onset (≤6 years), and <4 years duration of JIA (12). JIA-U also has a female predominance, and an association with HLA-DR8 is a risk factor although this does not inform the screening schedule (13-15). Uveitis is diagnosed based on the presence of inflammatory features including cells in the anterior chamber (AC) defined by the SUN grading criteria as well as AC flare from protein leakage (7,16,17).
Monitoring
Following a diagnosis of JIA-U, continued ophthalmic monitoring for inflammation and disease complications remains critical. The frequency of visits is based on disease severity, ocular complications, and treatment (11,18). In addition, continuous and frequent monitoring is especially needed when tapering medications to gauge relapses or increases in uveitis activity. While tapering or discontinuing topical glucocorticoids, ACR/AF strongly recommends ophthalmic monitoring within 1 month of each change in topical glucocorticoids (11). If tapering or discontinuing systemic therapy, monitoring is strongly recommended within 2 months of changing the regimen. If the patient is on stable therapy, monitoring can be reduced to every 3 months.
Continued monitoring of intraocular pressure (IOP) is vital in patients with JIA-U because of the increased risk of glaucoma and ocular hypertension (6,19). Angeles-Han et al. (6) reported other ocular complications including cataract (31%), synechiae (31%), band keratopathy (25%), and cystoid macular edema (CME) (15%). Regular assessment of AC cells and new or worsening ocular complications can provide monitoring of visual damage as well as show efficacy of treatment. Risk factors for visual impairment include increased severity of JIA-U and uveitis onset preceding arthritis (15,20). A retrospective cohort study by Thorne et al. (21) showed posterior synechiae, AC flare ≥1+, and abnormal IOP at presentation were risk factors for vision loss in patients with JIA-U. In follow-up visits, AC cells of ≥0.5+ was associated with an increased risk of visual impairment and blindness. To minimize the occurrence of these ocular complications related to JIA-U, early screening, diagnosis, and treatment are needed for this patient population.
Pathogenesis
While there is evidence showing an association between JIA and uveitis, the initiating events of uveitis immunopathology are not well understood. A combination of genetic and environmental factors is thought to contribute to its occurrence. Studies have shown an association within the human leukocyte antigen (HLA) area and have looked at the function of the various HLA alleles on the development of JIA (22,23). Specifically, combinations of HLA-DRB1 genes in children with JIA may predispose them to uveitis development (24). In addition, the current hypothesis proposes that both adaptive (antigen-specific) and innate (non-specific) responses contribute to uveitis (25). Uveitis may be caused by a loss of tolerance to auto-antigens and the activation of T lymphocytes (26). CD4+ cells (Th1, Th2, Th17) and CD8+ cells may also play an important role in autoimmune uveitis (23).
Various factors are critical in the inflammatory process. TNF-α is synthesized by monocytes, neutrophils, mast cells, macrophages, and both natural killer and T cells, and it drives Th1 cell responses (27,28). Increased expression of TNF-α has also been shown in experimental autoimmune uveitis at peak levels of inflammation (29). IL-6 is a cytokine derived from macrophages, which can function in both a pro- and anti-inflammatory fashion. It has been shown to play a role in the differentiation and proliferation of T cells (28,30,31). Janus kinase (JAK) mediated pathways are also involved in the pathogenesis of several autoimmune diseases including uveitis (32). All of these are vital players in the perpetuation of inflammation, and therapies may target these specific factors to limit the inflammation and ocular sequelae from JIA-uveitis.
Treatment
Synthetic treatment
Early detection and treatment are necessary to optimize the visual outcomes of children with JIA-U. The goal of treatment is to achieve inactive uveitis or an AC cell grade of 0 (33). Topical glucocorticoids (e.g., prednisolone acetate 1% or difluprednate 0.05%) are the first line of treatment for anterior uveitis and are used in 90% of patients with JIA-U (34,35). There are, however, adverse effects associated with long-term glucocorticoid use such as ocular hypertension and development of cataract (36). Prednisolone acetate is preferred before difluprednate (Durezol) because of increased adverse effects of difluprednate (37). However, increased disease severity may prompt the use of difluprednate in some patients but requires close monitoring for IOP-related adverse events, which has been shown to be common in individuals receiving difluprednate (38).
Local triamcinolone acetonide (TA) injections have also demonstrated efficacy, but local periocular or intravitreal injections may require general anesthesia for pediatric patients and require repeated administration, leading to increased risk for glaucoma and cataract development (34). Longer duration implants have also been explored to reduce the administration of medication. Dexamethasone 0.7 mg intravitreal insert (Ozurdex, Allergan) and fluocinolone acetonide 0.59 mg surgical intravitreal implant (Retisert, Bausch and Lomb) have shown efficacy in cases of refractory JIA-U but are associated with an increase in IOP and cataract formation, particularly with the Retisert implant (39-41). In general, glucocorticoid injections and implants are not recommended in children with JIA-U. Oral glucocorticoids may be used as bridging therapy but not for prolonged use.
Because of the adverse effects associated with prolonged corticosteroid use, disease modifying anti-rheumatic drugs (DMARDS) are commonly prescribed to patients with JIA-U. Methotrexate (10–25 mg/m2) administered orally or via subcutaneous injection is the most common first-line systemic agent, which is prescribed for 76% of patients with JIA-U (34,35,42). Methotrexate is an anti-metabolite that inhibits DNA replication and RNA transcription in B and T lymphocytes (43,44). Common adverse effects include gastrointestinal (GI) toxicity (oral ulcers, nausea, vomiting) and hepatorenal toxicity, so patients taking methotrexate should have regular lab monitoring for disease toxicity (34). Subcutaneous administration of methotrexate has a higher bioavailability and may minimize gastrointestinal side effects (42).
Other synthetic DMARDS are mycophenolate mofetil (MMF) and cyclosporine A (CsA) although these are prescribed less frequently for JIA-U as they are less effective for arthritis (45,46). MMF has similar GI toxicity effects as methotrexate in addition to hair loss and leukopenia. CsA, a T-cell calcineurin inhibitor, has been shown to cause nephrotoxicity and hirsutism as well as similar GI toxicity and hepatotoxicity as methotrexate (34), but it is less frequently prescribed in clinical practice (47).
Biologic treatment
TNF-α inhibitors
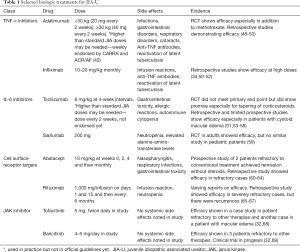
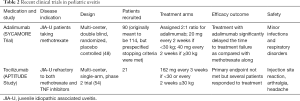
Biologic therapies are effective for children with JIA-U that is severe or refractory to methotrexate. TNF-α has been targeted to control the inflammatory response in JIA-U (Table 1), and adalimumab is a common anti-TNF-α prescribed for JIA-U refractory to methotrexate or severe in presentation. The SYCAMORE trial (Table 2) is a multicenter, double blind, randomized, placebo-controlled trial analyzing the efficacy of adalimumab with methotrexate for JIA-U (48). The standard dose of methotrexate given was 10 to 20 mg per square meter with a maximum dose of 25 mg. The subcutaneous adalimumab dose was 20 mg in patients weighing <30 kg or 40 mg in patients weighing ≥30 kg. During an interim analysis, adalimumab with methotrexate showed a significantly lower risk of treatment failure than the placebo-control group, and the trial recruitment was held due to these efficacy signals. However, adalimumab was associated with an increase in adverse effects compared to the control group, with some of these adverse effects including infections, gastrointestinal disorders, and respiratory disorders. A meta-analysis conducted by Jari et al. assessed the efficacy of adalimumab in 1,289 patients, and the pooled response rate of adalimumab was estimated to be 68.0% (95% CI: 65.4 to 70.6). Common side effects recorded in this meta-analysis included local pain, anemia, cataracts, and uveitis flares (49).
Full table
Full table
Infliximab is an anti-TNF-α treatment that is also commonly utilized for JIA-U. Infliximab has demonstrated efficacy for JIA-U when administered at high dosages (10–20 mg/kg/dose) with good tolerability (51,52). However, there has been variability in the dosing of infliximab and limited studies analyzing efficacy and safety, which may lead to differences in outcomes seen (34). In a retrospective study by Lerman et al. the efficacy of infliximab, adalimumab and etanercept were analyzed to note if minimal or no uveitis activity could be obtained with these treatments (50). Participants treated with infliximab had a median starting dose of 9.3 mg/kg (range 4.5 to 13.3 mg/kg) with only 5 subjects receiving treatment with doses <7 mg/kg. All participants treated with adalimumab received 40 mg every other week. Participants on etanercept received between 12.5–25 mg twice weekly, except for one small young child who received 0.8 mg/kg weekly. Treatment success was observed in 75% of patients overall, and there were better outcomes if diagnosed with JIA. In addition, adalimumab and infliximab were shown to be more effective than etanercept. A meta-analysis of anti-TNF-α on efficacy of childhood chronic uveitis has demonstrated superior efficacy with adalimumab and infliximab compared to etanercept, with the assertion that current evidence does not support the use of etanercept for JIA-U although evidence is limited (41,70,71).
IL-6 targets
IL-6 inhibitors can be considered for patients refractory to anti-TNF-α treatments (Table 1). These therapies are shown to be effective for rheumatoid arthritis especially since levels of IL-6 have been shown to correlate to disease severity (72,73). Based on this, the efficacy of IL-6 targets to treat JIA-U has been explored. Tocilizumab, a monoclonal antibody inhibitor of IL-6 receptor, is approved for treatment of JIA, and the standard dose is 8 mg/kg at 4-week intervals. Retrospective and limited prospective studies have shown efficacy in uveitis (31,53-55). Tocilizumab has been shown especially effective in pediatric patients with CME who are otherwise refractory to treatment (56,57). A multicenter, Phase 2 trial (APTITUDE study) (Table 2) assessed the efficacy of tocilizumab in 21 children with JIA-U who were refractory to methotrexate and TNF-α inhibitors (54). Participants continued a stable dose of methotrexate throughout the trial. Those weighing ≥30 kg were dosed with 162 mg of tocilizumab every 2 weeks while participants weighing <30 kg were dosed with 162 mg of tocilizumab every 3 weeks through a subcutaneous injection. The study did not meet its primary endpoint as 7 patients showed a response, but it did show promise as corticosteroid drops were tapered or stopped in several patients and resolution of three out of the four macular edema cases did occur. Adverse effects associated with administration of tocilizumab include autoimmune cytopenia, gastrointestinal toxicity, allergic reactions, and increased infections (55,58).
Another monoclonal antibody IL-6 inhibitor that shows potential in non-infectious uveitis is sarilumab. This therapy did show improvement in patients with non-infectious uveitis in the SATURN study, a Phase 2 study analyzing the efficacy and safety of sarilumab in adults. In this study, patients received treatment every 2 weeks for 16 weeks with subcutaneous sarilumab 200 mg or placebo. However, no similar studies have been conducted focusing on JIA-U (59). Side effects for sarilumab include neutropenia and elevated alanine-amino-transferase levels. Other IL-6 inhibitors are on the market; however, these have not been studied yet in terms of JIA-U or non-infectious uveitis.
Cell surface receptor targets
Abatacept (Table 1) is another biologic therapy that decreases CD80/CD86 expression on B-cells and prevents the activation of T cells. The standard dose is 10 mg/kg at weeks 0, 2, and 4 and then monthly (74). A prospective study of two patients with JIA-U who were refractory to maximum conventional treatment received IV abatacept at 10 mg/kg monthly. After 9 months, both patients remained in remission without steroids, but joint disease remained active in one of the cases. Abatacept is a promising treatment for refractory cases of JIA-uveitis, but concomitant therapies may be necessary to control the articular disease (60,61). A multinational retrospective study comparing abatacept as a first-line treatment and as a rescue treatment showed that the use of abatacept as a first line therapy was comparable to rescue treatment in refractory JIA-U (62). Side effects associated with abatacept can include nasopharyngitis, respiratory infections, and gastrointestinal toxicity (nausea), and in some studies, an exacerbation of the JIA-U has been seen with the use of abatacept (63,64).
Rituximab (Table 1) targets the CD20 antigen of B cells, which help promote differentiation of T regulatory cells (75). Standard dose of rituximab is a 1,000 mg/infusion on days 1 and 15 and then every 6 months (74). A retrospective multicenter study analyzed 10 patients with severe refractory JIA-U (65). Uveitis did improve in seven patients given rituximab (375 mg/m2 body surface) and allowed the tapering of glucocorticoids and DMARDs. However, new uveitis occurred in 4 of the 7 after 6–9 months. These recurrences were attributed to the survival of long-lived autoreactive plasma cells without the CD20 antigen because of B-cell depletion (65,66). A study by Miserocchi showed the efficacy of rituximab in eight patients who were refractory to three different TNF-α inhibitors with seven of them achieving persistent remission with tapered down cDMARDs (67). In this study, rituximab was given at the dose of 1,000 mg per infusion on days 1 and 15. While rituximab does show promise for treatment of refractory JIA-U, there have been varying reports on efficacy, and further studies are needed.
Janus kinase inhibitors (JAK inhibitors)
Despite a broad array of therapeutics available, some patients do not attain remission of their uveitis, and JAK inhibitors (Table 1) have emerged as another class of immunomodulatory therapies for these individuals. While evidence of JAK inhibition in the treatment of JIA-U is limited, Miserocchi et al. describes the use of JAK inhibitor therapies in four children with JIA-U refractory to other therapies such as infliximab, adalimumab, rituximab, abatacept and tocilizumab (32). Three cases were treated with baricitinib (4–5 mg/day) while one case used tofacitinib (5 mg, twice daily) as a therapy. All patients showed improvement of uveitis activity and control of intraocular inflammation; however, articular disease did not respond as favorably as the ophthalmic inflammation. In addition, the treatment was well tolerated in patients, and no systemic side effects, lab anomalies, or infections were noted in the 7-month follow-up period. Another report showed a positive effect of tofacitinib (5 mg, twice daily) in a patient with JIA-U and refractory macular edema (ME) (68). The role of JAK inhibition for refractory cases of JIA-U is unknown but requires further investigation. Currently, there is a clinical trial studying the efficacy of baricitinib in pediatric patients with JIA-U 69).
Relapse and remission
In recent years, there has been increased interest in understanding relapse and remission rates following discontinuation of immunomodulatory therapies (IMT). A retrospective case series found that 69% (8/13) patients treated with methotrexate relapsed after discontinuation of therapy. Predictors of relapse free survival according to this study included patient’s age >8 at withdrawal and inactivity of uveitis >2 years prior to withdrawal (76). Another study found that patients treated with IMT at an older age and later in their disease course had higher rates of relapse, often within one year of medication discontinuation (77).
A retrospective, multicenter, cohort study by Simonini et al. showed time to achieve inactivity was predictive of remission success in patients with JIA-U (78). Also, achieving inactivity and remission by anti-TNF-α compared to methotrexate therapies was predictive of a lower chance of remission after systemic treatment was discontinued. While another study showed a longer period of inactivity prior to withdrawal of methotrexate reduced the chances of relapse, this was not replicated by Simonini et al. (76,78).
A retrospective study conducted by Acharya et al. analyzed the relapse rates after discontinuation of IMT especially in cases of TNF-α inhibitors (79). Patients were separated based on their reason for discontinuation delineating those who stopped due to presumed remission versus other reasons. The study found that while corticosteroid-sparing treatment was achieved in a majority of the patients, the attempts to discontinue IMT were often unsuccessful. This study raised additional key questions regarding the appropriate timing of medication discontinuation and relapse rate. These questions will be assessed in the National Eye Institute-funded multicenter Adalimumab in JIA-associated Uveitis Stopping Trial, or ADJUST Study, a Phase 3 clinical trial analyzing the feasibility of discontinuing adalimumab in patients with JIA-U (80).
Conclusions
Uveitis is a common manifestation of JIA-U, but ocular complications that lead to vision loss may be prevented with regular screening and timely, effective therapy. JIA-U typically presents asymptomatically, thus early scheduled screening is critical. Prompt treatment and control of inflammation can improve visual outcomes and prevent vision loss. An improved understanding of the growing array of medications is needed for appropriate therapeutic choice. Methotrexate is the first-line corticosteroid-sparing treatment, but refractory uveitis, severe disease at onset, side effects, and intolerance may preclude its use in some patients.
Biologic therapies targeting inflammatory mechanisms are alternative treatments, which are increasingly used in clinical practice. TNF-α inhibitors such as adalimumab and infliximab are often the initial choices for biologic therapy for children with severe uveitis at onset or methotrexate failure. In patients refractory to anti-TNF-α factors, other biologics such as tocilizumab and abatacept may also be considered. In addition, emerging therapies targeting cell surface markers involved in the inflammatory cascade show promise especially for refractory cases but requires additional investigation. Further identification and study of inflammatory biomarker profiles are needed to develop targeted anti-inflammatory measures for patients with JIA-U.
Acknowledgments
Funding: This project was supported by unrestricted departmental grant from Research to Prevent Blindness, Inc. to the Emory Eye Center, Emory University School of Medicine, National Eye Institute/National Institutes of Health core grant P30-EY06360 (Department of Ophthalmology, Emory University School of Medicine), National Eye Institute of the National Institutes of Health under award number K23 EY030158 (Shantha), R01EY030521 (STAH), and R01 EY029594 (SY). The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health or the views or policies of the Department of Health and Human Services, nor does mention of trade names, commercial products, or organizations imply endorsement by the U.S. Government.
Footnote
Provenance and Peer Review: This article was commissioned by the Editorial Office, Annals of Eye Science for the series “Innovations in the Diagnosis and Management of Uveitis”. The article has undergone external peer review.
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/aes-2019-dmu-10). The series “Innovations in the Diagnosis and Management of Uveitis” was commissioned by the editorial office without any funding or sponsorship. Dr. SY served as the unpaid Guest Editor of the series. Dr. STAH reports a patent Effects of Youngsters’ Eyesight on QOL questionnaire licensed to Emory University. The authors have no other conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Petty RE, Southwood TR, Manners P, et al. International League of Associations for Rheumatology classification of juvenile idiopathic arthritis: second revision, Edmonton, 2001. J Rheumatol 2004;31:390-2. [PubMed]
- Denniston AK, Holland GN, Kidess A, et al. Heterogeneity of primary outcome measures used in clinical trials of treatments for intermediate, posterior, and panuveitis. Orphanet J Rare Dis 2015;10:97. [Crossref] [PubMed]
- Smith JA, Mackensen F, Sen HN, et al. Epidemiology and course of disease in childhood uveitis. Ophthalmology 2009;116:1544-51, 51.e1.
- Nordal E, Rypdal V, Christoffersen T, et al. Incidence and predictors of Uveitis in juvenile idiopathic arthritis in a Nordic long-term cohort study. Pediatr Rheumatol Online J 2017;15:66. [Crossref] [PubMed]
- Heiligenhaus A, Minden K, Föll D, et al. Uveitis in juvenile idiopathic arthritis. Dtsch Arztebl Int 2015;112:92-100. i. [PubMed]
- Angeles-Han ST, McCracken C, Yeh S, et al. Characteristics of a cohort of children with Juvenile Idiopathic Arthritis and JIA-associated Uveitis. Pediatr Rheumatol Online J 2015;13:19. [Crossref] [PubMed]
- Jabs DA, Nussenblatt RB, Rosenbaum JT. Standardization of uveitis nomenclature for reporting clinical data. Results of the First International Workshop. Am J Ophthalmol 2005;140:509-16. [Crossref] [PubMed]
- Foster CS. Diagnosis and treatment of juvenile idiopathic arthritis-associated uveitis. Curr Opin Ophthalmol 2003;14:395-8. [Crossref] [PubMed]
- Clarke SL, Sen ES, Ramanan AV. Juvenile idiopathic arthritis-associated uveitis. Pediatr Rheumatol Online J 2016;14:27. [Crossref] [PubMed]
- Guney ETT, I. Symptoms and Signs of Anterior Uveitis. US Ophthalmology Review 2013;6:33-7.
- Angeles-Han ST, Ringold S, Beukelman T, et al. 2019 American College of Rheumatology/Arthritis Foundation Guideline for the Screening, Monitoring, and Treatment of Juvenile Idiopathic Arthritis-Associated Uveitis. Arthritis Care Res (Hoboken) 2019;71:703-16. [Crossref] [PubMed]
- American Academy of Pediatrics Section on Rheumatology and Section on Ophthalmology. Guidelines for ophthalmologic examinations in children with juvenile rheumatoid arthritis. Pediatrics 1993;92:295-6. [PubMed]
- Martini A. Are the number of joints involved or the presence of psoriasis still useful tools to identify homogeneous disease entities in juvenile idiopathic arthritis? J Rheumatol 2003;30:1900-3. [PubMed]
- Zeggini E, Packham J, Donn R, et al. Association of HLA-DRB1*13 with susceptibility to uveitis in juvenile idiopathic arthritis in two independent data sets. Rheumatology (Oxford) 2006;45:972-4. [Crossref] [PubMed]
- Edelsten C, Lee V, Bentley CR, et al. An evaluation of baseline risk factors predicting severity in juvenile idiopathic arthritis associated uveitis and other chronic anterior uveitis in early childhood. Br J Ophthalmol 2002;86:51-6. [Crossref] [PubMed]
- Li Y, Lowder C, Zhang X, et al. Anterior chamber cell grading by optical coherence tomography. Invest Ophthalmol Vis Sci 2013;54:258-65. [Crossref] [PubMed]
- Agrawal RV, Murthy S, Sangwan V, et al. Current approach in diagnosis and management of anterior uveitis. Indian J Ophthalmol 2010;58:11-9. [Crossref] [PubMed]
- Constantin T, Foeldvari I, Anton J, et al. Consensus-based recommendations for the management of uveitis associated with juvenile idiopathic arthritis: the SHARE initiative. Ann Rheum Dis 2018;77:1107-17. [Crossref] [PubMed]
- Heinz C, Schumacher C, Roesel M, Heiligenhaus A. Elevated intraocular pressure in uveitis associated with juvenile idiopathic arthritis-associated uveitis, often detected after achieving inactivity. Br J Ophthalmol 2012;96:140-1. [Crossref] [PubMed]
- Kump LI, Castañeda RA, Androudi SN, et al. Visual outcomes in children with juvenile idiopathic arthritis-associated uveitis. Ophthalmology 2006;113:1874-7. [Crossref] [PubMed]
- Thorne JE, Woreta F, Kedhar SR, et al. Juvenile idiopathic arthritis-associated uveitis: incidence of ocular complications and visual acuity loss. Am J Ophthalmol 2007;143:840-6. [Crossref] [PubMed]
- Murray KJ, Moroldo MB, Donnelly P, et al. Age-specific effects of juvenile rheumatoid arthritis-associated HLA alleles. Arthritis Rheum 1999;42:1843-53. [Crossref] [PubMed]
- Kalinina Ayuso V, Makhotkina N, van Tent-Hoeve M, et al. Pathogenesis of juvenile idiopathic arthritis associated uveitis: the known and unknown. Surv Ophthalmol 2014;59:517-31. [Crossref] [PubMed]
- Angeles-Han ST, McCracken C, Yeh S, et al. HLA Associations in a Cohort of Children With Juvenile Idiopathic Arthritis With and Without Uveitis. Invest Ophthalmol Vis Sci 2015;56:6043-8. [Crossref] [PubMed]
- Lee RW, Dick AD. Current concepts and future directions in the pathogenesis and treatment of non-infectious intraocular inflammation. Eye (Lond) 2012;26:17-28. [Crossref] [PubMed]
- Caspi RR. A look at autoimmunity and inflammation in the eye. J Clin Invest 2010;120:3073-83. [Crossref] [PubMed]
- Dick AD, Forrester JV, Liversidge J, et al. The role of tumour necrosis factor (TNF-alpha) in experimental autoimmune uveoretinitis (EAU). Prog Retin Eye Res 2004;23:617-37. [Crossref] [PubMed]
- Ooi KG, Galatowicz G, Calder VL, et al. Cytokines and chemokines in uveitis: is there a correlation with clinical phenotype? Clin Med Res 2006;4:294-309. [Crossref] [PubMed]
- Okada AA, Sakai J, Usui M, et al. Intraocular cytokine quantification of experimental autoimmune uveoretinitis in rats. Ocul Immunol Inflamm 1998;6:111-20. [Crossref] [PubMed]
- La Flamme AC, Pearce EJ. The absence of IL-6 does not affect Th2 cell development in vivo, but does lead to impaired proliferation, IL-2 receptor expression, and B cell responses. J Immunol 1999;162:5829-37. [PubMed]
- Karkhur S, Hasanreisoglu M, Vigil E, et al. Interleukin-6 inhibition in the management of non-infectious uveitis and beyond. J Ophthalmic Inflamm Infect 2019;9:17. [Crossref] [PubMed]
- Miserocchi E, Giuffrè C, Cornalba M, et al. JAK inhibitors in refractory juvenile idiopathic arthritis-associated uveitis. Clin Rheumatol 2020;39:847-51. [Crossref] [PubMed]
- Bou R, Adán A, Borrás F, et al. Clinical management algorithm of uveitis associated with juvenile idiopathic arthritis: interdisciplinary panel consensus. Rheumatol Int 2015;35:777-85. [Crossref] [PubMed]
- Sood AB, Angeles-Han ST. An Update on Treatment of Pediatric Chronic Non-Infectious Uveitis. Curr Treatm Opt Rheumatol 2017;3:1-16. [Crossref] [PubMed]
- Henderson LA, Zurakowski D, Angeles-Han ST, et al. Medication use in juvenile uveitis patients enrolled in the Childhood Arthritis and Rheumatology Research Alliance Registry. Pediatr Rheumatol Online J 2016;14:9. [Crossref] [PubMed]
- Thorne JE, Woreta FA, Dunn JP, et al. Risk of cataract development among children with juvenile idiopathic arthritis-related uveitis treated with topical corticosteroids. Ophthalmology 2010;117:1436-41. [Crossref] [PubMed]
- Birnbaum AD, Jiang Y, Tessler HH, et al. Elevation of intraocular pressure in patients with uveitis treated with topical difluprednate. Arch Ophthalmol 2011;129:667-8. [Crossref] [PubMed]
- Slabaugh MA, Herlihy E, Ongchin S, et al. Efficacy and potential complications of difluprednate use for pediatric uveitis. Am J Ophthalmol 2012;153:932-8. [Crossref] [PubMed]
- Mehta PJ, Alexander JL, Sen HN. Pediatric uveitis: new and future treatments. Curr Opin Ophthalmol 2013;24:453-62. [Crossref] [PubMed]
- Leinonen S, Immonen I, Kotaniemi K. Fluocinolone acetonide intravitreal implant (Retisert(®)) in the treatment of sight threatening macular oedema of juvenile idiopathic arthritis-related uveitis. Acta Ophthalmol 2018;96:648-51. [Crossref] [PubMed]
- Rosenbaum JT, Bodaghi B, Couto C, et al. New observations and emerging ideas in diagnosis and management of non-infectious uveitis: A review. Semin Arthritis Rheum 2019;49:438-45. [Crossref] [PubMed]
- Angeles-Han ST, Lo MS, Henderson LA, et al. Childhood Arthritis and Rheumatology Research Alliance Consensus Treatment Plans for Juvenile Idiopathic Arthritis-Associated and Idiopathic Chronic Anterior Uveitis. Arthritis Care Res (Hoboken) 2019;71:482-91. [Crossref] [PubMed]
- Simonini G, Cantarini L, Bresci C, et al. Current therapeutic approaches to autoimmune chronic uveitis in children. Autoimmun Rev 2010;9:674-83. [Crossref] [PubMed]
- Simonini G, Paudyal P, Jones GT, et al. Current evidence of methotrexate efficacy in childhood chronic uveitis: a systematic review and meta-analysis approach. Rheumatology (Oxford) 2013;52:825-31. [Crossref] [PubMed]
- Doycheva D, Deuter C, Stuebiger N, et al. Mycophenolate mofetil in the treatment of uveitis in children. Br J Ophthalmol 2007;91:180-4. [Crossref] [PubMed]
- Kilmartin DJ, Forrester JV, Dick AD. Cyclosporin A therapy in refractory non-infectious childhood uveitis. Br J Ophthalmol 1998;82:737-42. [Crossref] [PubMed]
- Gerloni V, Cimaz R, Gattinara M, et al. Efficacy and safety profile of cyclosporin A in the treatment of juvenile chronic (idiopathic) arthritis. Results of a 10-year prospective study. Rheumatology (Oxford) 2001;40:907-13. [Crossref] [PubMed]
- Ramanan AV, Dick AD, Benton D, et al. A randomised controlled trial of the clinical effectiveness, safety and cost-effectiveness of adalimumab in combination with methotrexate for the treatment of juvenile idiopathic arthritis associated uveitis (SYCAMORE Trial). Trials 2014;15:14. [Crossref] [PubMed]
- Jari M, Shiari R, Salehpour O, et al. Epidemiological and advanced therapeutic approaches to treatment of uveitis in pediatric rheumatic diseases: a systematic review and meta-analysis. Orphanet J Rare Dis 2020;15:41. [Crossref] [PubMed]
- Lerman MA, Burnham JM, Chang PY, et al. Response of pediatric uveitis to tumor necrosis factor-α inhibitors. J Rheumatol 2013;40:1394-403. [Crossref] [PubMed]
- Kahn P, Weiss M, Imundo LF, et al. Favorable response to high-dose infliximab for refractory childhood uveitis. Ophthalmology 2006;113:860-4.e2. [Crossref] [PubMed]
- Sukumaran S, Marzan K, Shaham B, et al. High dose infliximab in the treatment of refractory uveitis: does dose matter? ISRN Rheumatol 2012;2012:765380 [Crossref] [PubMed]
- Tappeiner C, Heinz C, Ganser G, et al. Is tocilizumab an effective option for treatment of refractory uveitis associated with juvenile idiopathic arthritis? J Rheumatol 2012;39:1294-5. [Crossref] [PubMed]
- Ramanan AV, Dick AD, Guly C, et al. Tocilizumab in patients with anti-TNF refractory juvenile idiopathic arthritis-associated uveitis (APTITUDE): a multicentre, single-arm, phase 2 trial. Lancet Rheumatol 2020;2:e135-e141. [Crossref] [PubMed]
- Calvo-Río V, Santos-Gómez M, Calvo I, et al. Anti-Interleukin-6 Receptor Tocilizumab for Severe Juvenile Idiopathic Arthritis-Associated Uveitis Refractory to Anti-Tumor Necrosis Factor Therapy: A Multicenter Study of Twenty-Five Patients. Arthritis Rheumatol 2017;69:668-75. [Crossref] [PubMed]
- Adán A, Mesquida M, Llorenç V, et al. Tocilizumab treatment for refractory uveitis-related cystoid macular edema. Graefes Arch Clin Exp Ophthalmol 2013;251:2627-32. [Crossref] [PubMed]
- Vegas-Revenga N, Calvo-Río V, Mesquida M, et al. Anti-IL6-Receptor Tocilizumab in Refractory and Noninfectious Uveitic Cystoid Macular Edema: Multicenter Study of 25 Patients. Am J Ophthalmol 2019;200:85-94. [Crossref] [PubMed]
- Lopalco G, Fabiani C, Sota J, et al. IL-6 blockade in the management of non-infectious uveitis. Clin Rheumatol 2017;36:1459-69. [Crossref] [PubMed]
- Heissigerová J, Callanan D, de Smet MD, Srivastava SK, et al. Efficacy and Safety of Sarilumab for the Treatment of Posterior Segment Noninfectious Uveitis (SARIL-NIU):: The Phase 2 SATURN Study. Ophthalmology 2019;126:428-37. [Crossref] [PubMed]
- Kenawy N, Cleary G, Mewar D, et al. Abatacept: a potential therapy in refractory cases of juvenile idiopathic arthritis-associated uveitis. Graefes Arch Clin Exp Ophthalmol 2011;249:297-300. [Crossref] [PubMed]
- Zulian F, Balzarin M, Falcini F, et al. Abatacept for severe anti-tumor necrosis factor alpha refractory juvenile idiopathic arthritis-related uveitis. Arthritis Care Res (Hoboken) 2010;62:821-5. [Crossref] [PubMed]
- Birolo C, Zannin ME, Arsenyeva S, et al. Comparable Efficacy of Abatacept Used as First-line or Second-line Biological Agent for Severe Juvenile Idiopathic Arthritis-related Uveitis. J Rheumatol 2016;43:2068-73. [Crossref] [PubMed]
- Tappeiner C, Miserocchi E, Bodaghi B, et al. Abatacept in the treatment of severe, longstanding, and refractory uveitis associated with juvenile idiopathic arthritis. J Rheumatol 2015;42:706-11. [Crossref] [PubMed]
- Gaggiano C, Rigante D, Tosi GM, et al. Treating juvenile idiopathic arthritis (JIA)-related uveitis beyond TNF-α inhibition: a narrative review. Clin Rheumatol 2020;39:327-37. [Crossref] [PubMed]
- Heiligenhaus A, Miserocchi E, Heinz C, et al. Treatment of severe uveitis associated with juvenile idiopathic arthritis with anti-CD20 monoclonal antibody (rituximab). Rheumatology (Oxford) 2011;50:1390-4. [Crossref] [PubMed]
- Mahévas M, Michel M, Weill JC, et al. Long-lived plasma cells in autoimmunity: lessons from B-cell depleting therapy. Front Immunol 2013;4:494. [Crossref] [PubMed]
- Miserocchi E, Pontikaki I, Modorati G, et al. Rituximab for uveitis. Ophthalmology 2011;118:223-4. [Crossref] [PubMed]
- Bauermann P, Heiligenhaus A, Heinz C. Effect of Janus Kinase Inhibitor Treatment on Anterior Uveitis and Associated Macular Edema in an Adult Patient with Juvenile Idiopathic Arthritis. Ocul Immunol Inflamm 2019;27:1232-4. [Crossref] [PubMed]
- A Study of Baricitinib (LY3009104) in Participants From 2 Years to Less Than 18 Years Old With Active JIA-Associated Uveitis or Chronic Anterior Antinuclear Antibody-Positive Uveitis. Available online: https://ClinicalTrials.gov/show/NCT04088409.
- Simonini G, Druce K, Cimaz R, et al. Current evidence of anti-tumor necrosis factor α treatment efficacy in childhood chronic uveitis: a systematic review and meta-analysis approach of individual drugs. Arthritis Care Res (Hoboken) 2014;66:1073-84. [Crossref] [PubMed]
- Smith JA, Thompson DJ, Whitcup SM, et al. A randomized, placebo-controlled, double-masked clinical trial of etanercept for the treatment of uveitis associated with juvenile idiopathic arthritis. Arthritis Rheum 2005;53:18-23. [Crossref] [PubMed]
- Srirangan S, Choy EH. The role of interleukin 6 in the pathophysiology of rheumatoid arthritis. Ther Adv Musculoskelet Dis 2010;2:247-56. [Crossref] [PubMed]
- Ogata A, Kato Y, Higa S, et al. IL-6 inhibitor for the treatment of rheumatoid arthritis: A comprehensive review. Mod Rheumatol 2019;29:258-67. [Crossref] [PubMed]
- Kim L, Li A, Angeles-Han S, et al. Update on the management of uveitis in children: an overview for the clinician. Expert review of ophthalmology 2019;14:211-8. [Crossref] [PubMed]
- Catzola V, Battaglia A, Buzzonetti A, et al. Changes in regulatory T cells after rituximab in two patients with refractory myasthenia gravis. J Neurol 2013;260:2163-5. [Crossref] [PubMed]
- Kalinina Ayuso V, van de Winkel EL, Rothova A, et al. Relapse rate of uveitis post-methotrexate treatment in juvenile idiopathic arthritis. Am J Ophthalmol 2011;151:217-22. [Crossref] [PubMed]
- Saboo US, Metzinger JL, Radwan A, et al. Risk factors associated with the relapse of uveitis in patients with juvenile idiopathic arthritis: a preliminary report. J aapos 2013;17:460-4. [Crossref] [PubMed]
- Simonini G, Bracaglia C, Cattalini M, et al. Predictors of Relapse after Discontinuing Systemic Treatment in Childhood Autoimmune Chronic Uveitis. J Rheumatol 2017;44:822-6. [Crossref] [PubMed]
- Acharya NR, Patel S, Homayounfar G, et al. Relapse of Juvenile Idiopathic Arthritis-Associated Uveitis after Discontinuation of Immunomodulatory Therapy. Ocul Immunol Inflamm 2019;27:686-92. [Crossref] [PubMed]
Adalimumab in JIA-associated Uveitis Stopping Trial
Cite this article as: Thomas J, Kuthyar S, Shantha JG, Angeles-Han ST, Yeh S. Update on biologic therapies for juvenile idiopathic arthritis-associated uveitis. Ann Eye Sci 2021;6:19.


