
Shunting, optic nerve sheath fenestration and dural venous stenting for medically refractory idiopathic intracranial hypertension: systematic review and meta-analysis
Introduction
Idiopathic intracranial hypertension (IIH) is a syndrome defined by increased intracranial pressure without ventriculomegaly or radiographic evidence of a mass lesion, and with normal cerebrospinal fluid (CSF) composition (1). First described by Quincke et al. in 1893 as “meningitis serosa”, IIH is a rare condition with only 0.9 cases per 100,00 in the general population (1-4). In comparison to the general population, IIH is most common in obese women aged between 20 and 44 years of age, with an overall prevalence of 15–19/100,000 in North America (2-4).
Given the lack of pathological or radiological evidence, IIH is a diagnosis of exclusion. Currently, IIH is defined by the Modified Dandy Criteria, which for diagnosis, requires signs and symptoms referable only to elevated intracranial pressure, a CSF opening pressure >25 cmH2O in the lateral decubitus position with normal CSF composition and no evidence of an underlying structural cause on imaging (1). Symptomatically, headache is the presenting complaint in 92–94% of patients (5,6). However, IIH can also lead to papilloedema, which if untreated, can lead to permanent loss of vision (1,7-12). As such, treatment of IIH is of paramount importance.
Despite the pathogenesis of IIH remaining largely speculative, therapeutic developments have substantially advanced patient management (12). Medical management of IIH includes acetazolamide, diuretics, weight loss and serial high-volume lumbar punctures (13). Unfortunately, not all patients respond to the aforementioned medical treatments. In medically refractory IIH, a CSF-diversion procedure, including ventriculoperitoneal (VP) and lumboperitoneal (LP) shunts, and optic nerve sheath fenestration (ONSF) are the next line of treatment (14). Furthermore, cerebral venous sinus stenosis has increasingly been recognised as a cause of increased intracranial pressure. First described by Higgins et al. in 2002, dural venous sinus stenting has increasingly been reported to have favourable clinical outcomes in the management of IIH (9,11,15-20).
Traditionally, the treatment for medically refractive IIH has been a CSF-diversion procedure, however alternative techniques have since been developed and used. This article presents an up-to-date meta-analysis and comparison of CSF-diversion procedures, venous sinus stent placement and optic nerve sheath fenestration for the treatment of medically refractive IIH from 1988 to 2017. In order to comprehensively analyse the procedures, an in-depth comparison of all three treatments with respect to post-operative improvement in headache, papilloedema and visual acuity, complications and the requirement for repeat procedures is completed.
Methods
The present systematic review and meta-analysis was conducted according to recommended guidelines (21,22).
Literature search strategy
Electronic searches were performed using Ovid Medline, PubMed, Cochrane Central Register of Controlled Trials (CCTR), Cochrane Database of Systematic Reviews (CDSR), ACP Journal Club, and Database of Abstracts of Review of Effectiveness (DARE) from their dates of inception to January 2017. To achieve the maximum sensitivity of the search strategy, we combined the terms: “idiopathic intracranial hypertension”, “pseudotumor cerebri”, “benign intracranial hypertension”, “shunt”, “optic nerve sheath decompression”, and “venous stenting”, as either key words or MeSH terms. The reference lists of all retrieved articles were reviewed for further identification of potentially relevant studies and these were then assessed using the inclusion and exclusion criteria.
Selection criteria
Eligible studies for the present systematic review and meta-analysis included those in which patient cohorts underwent an index procedure for treatment of medically refractory IIH via shunting, optic nerve sheath fenestration or venous stenting. Cohorts focusing on patients with repeat procedures were excluded from analysis. Studies that did not include complications as endpoints were excluded. When institutions published duplicate studies with accumulating numbers of patients or increased lengths of follow-up, only the most complete reports were included for quantitative assessment at each time interval. All publications were limited to those involving human subjects and in the English language. Abstracts, case reports, conference presentations, editorials, reviews and expert opinions were excluded.
Data extraction
All data were extracted from article texts, tables and figures. Two investigators independently reviewed each retrieved article (K Phan, GT Nguyen). Discrepancies between the two reviewers were resolved by discussion and consensus.
Statistical analysis
A meta-analysis of proportions was conducted for the available perioperative and postoperative variables. Heterogeneity was evaluated using Cochran Q and I2 test. Weighted means were calculated by determining the total number of events divided by total sample size. Subgroup analysis was conducted using mixed-effects meta-regression with a fixed-effect moderator variable for the intervention. All analyses were performed using the “metafor” package for R version 3.02. P values <0.05 were considered statistically significant.
Results
A total of 1,498 references were identified from the electronic database searches. After exclusion and inclusion criteria were applied, a total of 55 studies were included in the analysis (Figure S1), including 22 shunt studies (10,23-43), 21 optic nerve fenestration studies (44-64), and 12 venous stenting studies (9,11,16-20,65-68). Baseline characteristics are summarized in Tables S1-3.
Post-operative improvements in headache, papilloedema and visual acuity, complication rates and the need for a subsequent procedure were analysed for CSF-diversion procedure, venous stent placement and optic nerve sheath fenestration.
CSF-diversion procedure
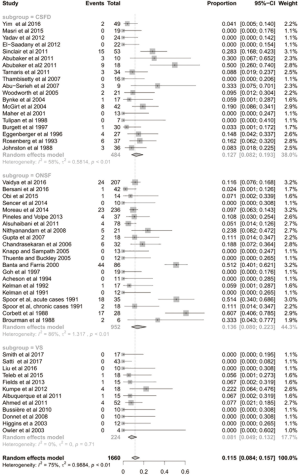
A total of 22 studies utilizing a CSF-diversion procedure met the inclusion criteria and were analysed (Table S1). This included 538 patients; 85% (398/466) were females. The mean age at presentation was 30.3 years. The mean BMI was 35.2 kg/m2 and the mean CSF opening pressure was 41.6 cmH2O.
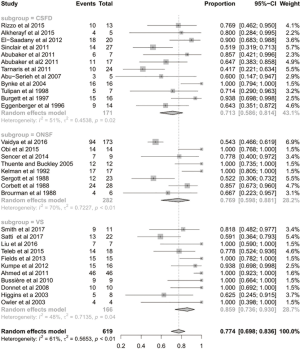
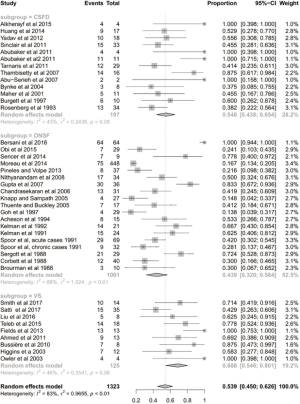
The mean follow-up time was 42 months. After the CSF-diversion procedure, 84% of patients had post-operative improvement in headache (95% CI: 0.688–0.923; I2=79%; Figure 1). Seventy-one percent of patients (95% CI: 0.586–0.814; I2=51%; Figure 2) and 55% of patients (95% CI: 0.438–0.654; I2=43%; Figure 3) had post-operative improvement in papilloedema and visual acuity, respectively, following the CSF-diversion procedure.
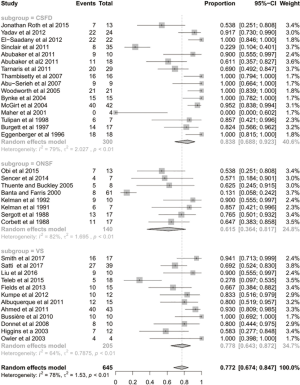
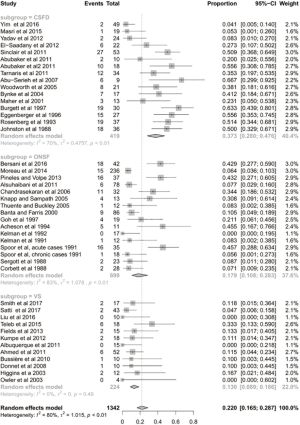
The complication rate, not including the need for a subsequent procedure, was 13% (95% CI: 0.082–0.193; I2=58%; Figure S2). The specific complications are listed in Table S1. Thirty-seven percent of patients undergoing a CSF-diversion procedure required a subsequent procedure (95% CI: 0.280–0.476; I2=70%; Figure 4). Specifically, 16 of the 22 studies analysed reported the requirement for a subsequent procedure, with 157 of the 538 patients undergoing an additional 540 procedures.
We also performed a subgroup analysis to compare patients with LP vs. VP shunt methods of CSF-diversion. We found no significant differences between LP vs. VP shunts in terms of improvement in headaches (87.4% vs. 88.2%), papilloedema (77.9% vs. 79%), visual acuity changes (51.8% vs. 49.9%), complication rate (8.1% vs. 15.7%) or subsequent procedure rate (34.5% vs. 47.6%).
Venous stent placement
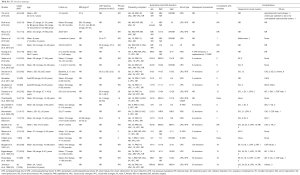
A total of 12 studies and 224 patients were included in the meta-analysis of dural venous stent placement (Table S2). Eighty-eight percent of patient (197/224) were female and the mean age was 33.4 years. The mean BMI was 34.8 kg/m2 and the mean CSF opening pressure was 36.1 cmH2O.
The mean follow-up time was 20 months. After dural venous stent placement, 78% of patients had post-operative improvement in headache (95% CI: 0.643–0.872; P<0.01; Figure 2). Eighty-six percent of patients (95% CI: 0.736–0.930; P=0.04; Figure 3) and 69% of patients (95% CI: 0.546–0.801; P=0.06; Figure 3) had post-operative improvement in papilloedema and visual acuity, respectively, following dural venous stent placement.
The complication rate, not including the need for a subsequent procedure, was 8% (95% CI: 0.049–0.132; P=0.71; Figure S2). Thirteen percent of patients undergoing venous stent placement required a subsequent procedure (95% CI: 0.089–0.186; P=0.49; Figure 4). All 12 of the included studies reported the requirement for subsequent procedures, which demonstrated that only 24 of 224 patients underwent additional procedures.
Optic nerve sheath fenestration
A total of 21 studies, including 872 patients and 1,455 eyes, met the inclusion criteria for the meta-analysis (Table S3). Forty-three percent of patients had a unilateral procedure while the remaining 57% of patients underwent bilateral ONSF. Eighty-three percent of patients (522/626) were females and the mean age was 32.2 years. The mean BMI was 33.5 kg/m2 and the mean CSF opening pressure was 33.2 cmH2O.
The mean follow-up time was 22 months. Following optic nerve sheath fenestration, 62% of patients had post-operative improvement in headache (95% CI: 0.364–0.817; P<0.01; Figure 1). Seventy-seven percent of patients (95% CI: 0.598–0.881; P<0.01; Figure 2) and 44% of patients (95% CI: 0.320–0.564; P<0.01; Figure 3) had post-operative improvement in papilloedema and visual acuity, respectively, following ONSF.
The complication rate, not including the need for a subsequent procedure, was 14% (95% CI: 0.080–0.223; P<0.01; Figure S2). Eighteen percent of patients undergoing ONSF required a subsequent procedure (95% CI: 0.108–0.283; P<0.01; Figure 4). Specifically, 16 of 21 studies reported the requirement for subsequent procedures with 111 patients requiring subsequent procedures out of 699 patients.
Comparison between treatment modalities for medically refractive IIH
When comparing CSF-diversion procedures, venous stent placement and ONSF with respect to improvement in headache and papilloedema post-operatively, the outcomes were not significantly different between the treatment modalities (P=0.223 and 0.192, respectively). However, post-operative improvement in visual acuity significantly favoured venous stent placement (P=0.037).
With respect to complication rates, no statistically significant differences were identified between the three treatment modalities (P=0.28), however, there was a significantly lower rate of subsequent procedures required following venous stent placement when compared to CSF-diversion procedures and ONSF (P<0.001). Leave-one-out sensitivity analysis did not significantly change the trend of the results.
Discussion
Characterised by increased intracranial pressure without a mass lesion or hydrocephalus, IIH classically presents with headache in obese women of childbearing age. Ophthalmologic signs, including diminished visual acuity and papilloedema on fundoscopic examination, frequently present alongside the headache (69). Given that papilloedema associated visual loss is a principle morbidity associated with the condition, the terms “benign intracranial hypertension” and “pseudotumour cerebri” no longer represent current nomenclature and in 2011, the term “idiopathic intracranial hypertension” was adopted (12,69).
Conservative therapy, including weight loss, repeated high-volume lumbar punctures and medications to reduce CSF production, such as acetazolamide, are the mainstays of treatment. However, some patients are non-responsive to medical therapy and as such, experience progressive worsening of symptoms or develop visual changes. For patients with medically refractive IIH, these patients are traditionally referred for ONSF or a CSF-diversion procedure including VP and LP shunting (70,71). However, more recently, dural venous sinus stent placement has been described in the literature as a safe and effective procedure for the management of medically refractive IIH.
CSF-diversion procedures appear to have the highest success rate for post-operative improvement in patient’s experiencing headaches. Eighty-four percent of patients who underwent a CSF-diversion procedure had post-operative improvement in headaches, compared to 79% and 62% for venous sinus stenting and ONSF, respectively. However, when all treatment modalities were compared, no statistically significant difference was detected (P=0.223).
Venous sinus stent placement was associated with the greatest post-operative improvement in both papilloedema and visual acuity with 86% and 69% of patients had improvements, respectively. This is in comparison to 77% and 44% of patients having post-operative improvement in papilloedema and visual acuity following ONSF, respectively. Seventy-one percent and 55% of patients had post-operative improvement in papilloedema and visual acuity following a CSF-diversion procedure, respectively. Despite the data favouring venous sinus stenting for patients with papilloedema, no statistically significant difference was detected between the treatment modalities (P=0.192). However, this is not the case with post-operative improvement in visual acuity, which showed a statistically significant difference favouring venous sinus stenting compared to CSF-diversion and ONSF (P=0.037).
Complication rates between the three treatment modalities were comparable. Optic nerve sheath fenestration had the greatest complication rate at 14%, which was comparable to the 13% complication rate for CSF-diversion procedures. Although the complication rate for venous sinus stenting was 8%, the lowest amongst the three treatment modalities, when compared against each other, no statistically significant difference was detected (P=0.2781).
Although the complication rate for CSF-diversion procedures is only 14%, 37% of patients undergoing such procedures require repeat or subsequent procedures. Of the 538 patients included in the meta-analysis, 157 patients underwent an additional 540 procedures, most of which were shunt revisions. With a mean follow-up time of 42 months, the high reported repeat procedure rate is concerning given that the mean age of patients undergoing a CSF-diversion procedure is 30.3 years. When compared to ONSF, which has a similar complication rate, the requirement for subsequent procedures is only 18%.
Venous sinus stent placement had a significantly lower revision rate of 13% when compared to CSF-diversion procedures and ONSF (P<0.001). Of the 224 patients who underwent venous sinus stenting included in the meta-analysis, only 24 patients required a subsequent procedure, of which, 12.5% had a CSF-diversion procedure. Although only 224 patients were included in the meta-analysis of dural venous sinus stent placement, the data suggests that this treatment modality is significantly more effective in improving visual acuity post-operatively and has a significantly lower requirement for additional procedures when compared to CSF-diversion procedures and ONSF.
Given the high complication rate and requirement for subsequent procedures, CSF-diversion procedures are associated with significant morbidity and cost. As such, the use of CSF-diversion procedures as the standard of treatment for medically refractive IIH should be reconsidered. When comparing the cost of venous sinus stent placement and CSF-diversion procedure, Ahmed et al. found that there was no significant cost difference between the insertion of an initial venous sinus stent and initial CSF shunt. (72) Although there is no significant cost difference with respect to the initial procedure, 87% of stents placed required only one stent procedure, in comparison to only 45% of shunts requiring only one shunt procedure (72). Given the high rate of repeat procedures, CSF-diversion procedures end up costing significantly more in the long term.
Although the results of this meta-analysis suggest the venous sinus stenting is a viable alternative to traditional CSF-diversion procedures and ONSF, there is still a limited, though growing, literature for this procedure in medically refractive IIH. The present analysis is constrained by several limitations. These include the lack of direct comparative analyses between studies and patient matching, as baseline patient differences could be confounders in the present analysis. One would expect that patients undergoing venous stenting procedures to have venous pathology, although the extent may vary between patients and studies. The retrospective nature of the included studies means the data is susceptible to selection bias. Given the relative low number of studies of venous sinus stenting when compared to that available for CSF-diversion procedures and ONSF, combined with the retrospective nature of a meta-analysis, confirmation of these results ideally requires a randomised controlled trial before one can confidently state which treatment modality is superior in the management of medically refractive IIH.
Conclusions
CSF-diversion procedures have traditionally been the standard of treatment for patients with medically refractive IIH. However, the results of our meta-analysis suggest that, with its lower complication rate, lower requirement for subsequent procedures and its superiority with respect to improving visual acuity, dural venous sinus stenting may be a viable alternative to traditional surgical interventions in patients refractory to medical treatment.
Full table

Full table

Full table
Acknowledgments
Funding: None.
Footnote
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/aes.2018.05.01). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Friedman DI, Jacobson DM. Diagnostic criteria for idiopathic intracranial hypertension. Neurology 2002;59:1492-5. [Crossref] [PubMed]
- Puffer RC, Mustafa W, Lanzino G. Venous sinus stenting for idiopathic intracranial hypertension: a review of the literature. J Neurointerv Surg 2013;5:483-6. [Crossref] [PubMed]
- Durcan PJ, Corbett JJ, Wall M. The incidence of pseudotumor cerebri: population studies in Iowa and Louisiana. Arch Neurol 1988;45:875-7. [Crossref] [PubMed]
- Radhakrishnan K, Ahlskog JE, Cross SA, et al. Idiopathic intracranial hypertension (pseudotumor cerebri): descriptive epidemiology in Rochester, Minn, 1976 to 1990. Arch Neurol 1993;50:78-80. [Crossref] [PubMed]
- Francis CE, Quiros PA. Headache management in idiopathic intracranial hypertension. Int Ophthalmol Clin 2014;54:103-14. [Crossref] [PubMed]
- Wall M. The headache profile of idiopathic intracranial hypertension. Cephalalgia 1990;10:331-5. [Crossref] [PubMed]
- Ball AK, Clarke CE. Idiopathic intracranial hypertension. Lancet Neurol 2006;5:433-42. [Crossref] [PubMed]
- Biousse V, Bruce BB, Newman NJ. Update on the pathophysiology and management of idiopathic intracranial hypertension. J Neurol Neurosurg Psychiatry 2012;83:488-94. [Crossref] [PubMed]
- Donnet A, Metellus P, Levrier O, et al. Endovascular treatment of idiopathic intracranial hypertension Clinical and radiologic outcome of 10 consecutive patients. Neurology 2008;70:641-7. [Crossref] [PubMed]
- Eggenberger ER, Miller NR, Vitale S. Lumboperitoneal shunt for the treatment of pseudotumor cerebri. Neurology 1996;46:1524-30. [Crossref] [PubMed]
- Fields JD, Javedani PP, Falardeau J, et al. Dural venous sinus angioplasty and stenting for the treatment of idiopathic intracranial hypertension. J Neurointerv Surg 2013;5:62-8. [Crossref] [PubMed]
- Friedman DI, Jacobson DM. Idiopathic intracranial hypertension. J Neuroophthalmol 2004;24:138-45. [Crossref] [PubMed]
- Satti S, Leishangthem L, Chaudry M. Meta-analysis of CSF diversion procedures and dural venous sinus stenting in the setting of medically refractory idiopathic intracranial hypertension. AJNR Am J Neuroradiol 2015;36:1899-904. [Crossref] [PubMed]
- Galgano MA, Deshaies EM. An update on the management of pseudotumor cerebri. Clin Neurol Neurosurg 2013;115:252-9. [Crossref] [PubMed]
- Higgins JNP, Owler BK, Cousins C, et al. Venous sinus stenting for refractory benign intracranial hypertension. Lancet 2002;359:228-30. [Crossref] [PubMed]
- Kumpe DA, Bennett JL, Seinfeld J, et al. Dural sinus stent placement for idiopathic intracranial hypertension. J Neurosurg 2012;116:538-48. [Crossref] [PubMed]
- Ahmed R, Wilkinson M, Parker G, et al. Transverse sinus stenting for idiopathic intracranial hypertension: a review of 52 patients and of model predictions. AJNR Am J Neuroradiol 2011;32:1408-14. [Crossref] [PubMed]
- Albuquerque FC, Dashti SR, Hu YC, et al. Intracranial venous sinus stenting for benign intracranial hypertension: clinical indications, technique, and preliminary results. World Neurosurg 2011;75:648-52. [Crossref] [PubMed]
- Higgins JN, Cousins C, Owler BK, et al. Idiopathic intracranial hypertension: 12 cases treated by venous sinus stenting. J Neurol Neurosurg Psychiatry 2003;74:1662-6. [Crossref] [PubMed]
- Bussière M, Falero R, Nicolle D, et al. Unilateral transverse sinus stenting of patients with idiopathic intracranial hypertension. AJNR Am J Neuroradiol 2010;31:645-50. [Crossref] [PubMed]
- Moher D, Liberati A, Tetzlaff J, et al. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med 2009;6:e1000097 [Crossref] [PubMed]
- Phan K, Mobbs RJ. Systematic reviews and meta-analyses in spine surgery, neurosurgery and orthopedics: guidelines for the surgeon scientist. J Spine Surg 2015;1:19-27. [PubMed]
- Yim B, Reid Gooch M, Dalfino JC, et al. Optimizing ventriculoperitoneal shunt placement in the treatment of idiopathic intracranial hypertension: an analysis of neuroendoscopy, frameless stereotaxy, and intraoperative CT. Neurosurg Focus 2016;40:E12 [Crossref] [PubMed]
- Roth J, Constantini S, Kesler A. Over-drainage and persistent shunt-dependency in patients with idiopathic intracranial hypertension treated with shunts and bariatric surgery. Surg Neurol Int 2015;6:S655-60. [Crossref] [PubMed]
- Rizzo JL, Lam KV, Wall M, et al. Perimetry, retinal nerve fiber layer thickness and papilledema grade after cerebrospinal fluid shunting in patients with idiopathic intracranial hypertension. J Neuroophthalmol 2015;35:22-5. [Crossref] [PubMed]
- Masri A, Jaafar A, Noman R, et al. Intracranial Hypertension in Children: Etiologies, Clinical Features, and Outcome. J Child Neurol 2015;30:1562-8. [Crossref] [PubMed]
- Alkherayf F, Abou Al-Shaar H, Awad M. Management of idiopathic intracranial hypertension with a programmable lumboperitoneal shunt: Early experience. Clin Neurol Neurosurg 2015;136:5-9. [Crossref] [PubMed]
- Huang LC, Winter TW, Herro AM, et al. Ventriculoperitoneal Shunt as a Treatment of Visual Loss in Idiopathic Intracranial Hypertension. J Neuroophthalmol 2014;34:223-8. [Crossref] [PubMed]
- Yadav YR, Parihar V, Agarwal M, et al. Lumbar peritoneal shunt in idiopathic intracranial hypertension. Turk Neurosurg 2012;22:21-6. [PubMed]
- El-Saadany WF, Farhoud A, Zidan I. Lumboperitoneal shunt for idiopathic intracranial hypertension: patients' selection and outcome. Neurosurg Rev 2012;35:239-43; discussion 243-4. [Crossref] [PubMed]
- Sinclair AJ, Kuruvath S, Sen D, et al. Is cerebrospinal fluid shunting in idiopathic intracranial hypertension worthwhile? A 10-year review. Cephalalgia 2011;31:1627-33. [Crossref] [PubMed]
- Abubaker K, Ali Z, Raza K, et al. Idiopathic intracranial hypertension: lumboperitoneal shunts versus ventriculoperitoneal shunts - case series and literature review. Br J Neurosurg 2011;25:94-9. [Crossref] [PubMed]
- Tarnaris A, Toma AK, Watkins LD, et al. Is there a difference in outcomes of patients with idiopathic intracranial hypertension with the choice of cerebrospinal fluid diversion site: A single centre experience. Clin Neurol Neurosurg 2011;113:477-9. [Crossref] [PubMed]
- Thambisetty M, Lavin PJ, Newman NJ, et al. Fulminant idiopathic intracranial hypertension. Neurology 2007;68:229-32. [Crossref] [PubMed]
- Abu-Serieh B, Ghassempour K, Duprez T, et al. Stereotactic ventriculoperitoneal shunting for refractory idiopathic intracranial hypertension. Neurosurgery 2007;60:1039-43. [Crossref] [PubMed]
- Woodworth GF, McGirt MJ, Elfert P, et al. Frameless stereotactic ventricular shunt placement for idiopathic intracranial hypertension. Stereotact Funct Neurosurg 2005;83:12-6. [Crossref] [PubMed]
- Bynke G, Zemack G, Bynke H, et al. Ventriculoperitoneal shunting for idiopathic intracranial hypertension. Neurology 2004;63:1314-6. [Crossref] [PubMed]
- McGirt MJ, Woodworth G, Thomas G, et al. Cerebrospinal fluid shunt placement for pseudotumor cerebri-associated intractable headache: predictors of treatment response and an analysis of long-term outcomes. J Neurosurg 2004;101:627-32. [Crossref] [PubMed]
- Maher CO, Garrity JA, Meyer FB. Refractory idiopathic intracranial hypertension treated with stereotactically planned ventriculoperitoneal shunt placement. Neurosurg Focus 2001;10:E1 [Crossref] [PubMed]
- Tulipan N, Lavin PJ, Copeland M. Stereotactic ventriculoperitoneal shunt for idiopathic intracranial hypertension: technical note. Neurosurgery 1998;43:175-6; discussion 176-7. [Crossref] [PubMed]
- Burgett RA, Purvin VA, Kawasaki A. Lumboperitoneal shunting for pseudotumor cerebri. Neurology 1997;49:734-9. [Crossref] [PubMed]
- Rosenberg ML, Corbett JJ, Smith C, et al. Cerebrospinal fluid diversion procedures in pseudotumor cerebri. Neurology 1993;43:1071-2. [Crossref] [PubMed]
- Johnston I, Besser M, Morgan MK. Cerebrospinal fluid diversion in the treatment of benign intracranial hypertension. J Neurosurg 1988;69:195-202. [Crossref] [PubMed]
- Vaidya NS, Mahmoud AM, Buzzacco D, et al. Visual outcomes following optic nerve sheath fenestration via the medial transconjunctival approach. Orbit 2016;35:271-7. [Crossref] [PubMed]
- Bersani TA, Meeker AR, Sismanis DN, et al. Pediatric and adult vision restoration after optic nerve sheath decompression for idiopathic intracranial hypertension. Orbit 2016;35:132-9. [Crossref] [PubMed]
- Obi EE, Lakhani BK, Burns J, et al. Optic nerve sheath fenestration for idiopathic intracranial hypertension: a seven year review of visual outcomes in a tertiary centre. Clin Neurol Neurosurg 2015;137:94-101. Erratum in: Corrigendum to "Optic nerve sheath fenestration for idiopathic intracranial hypertension: A seven year review of visual outcomes in a tertiary centre" Clin Neurol Neurosurg 2017;137:94-101. [Crossref] [PubMed]
- Sencer A, Akcakaya MO, Basaran B, et al. Unilateral endoscopic optic nerve decompression for idiopathic intracranial hypertension: a series of 10 patients. World Neurosurg 2014;82:745-50. [Crossref] [PubMed]
- Moreau A, Lao KC, Farris BK. Optic nerve sheath decompression: a surgical technique with minimal operative complications. J Neuroophthalmol 2014;34:34-8. [Crossref] [PubMed]
- Pineles SL, Volpe NJ. Long-Term Results of Optic Nerve Sheath Fenestration for Idiopathic Intracranial Hypertension: Earlier Intervention Favours Improved Outcomes. Neuroophthalmology 2013;37:12-9. [Crossref] [PubMed]
- Alsuhaibani AH, Carter KD, Nerad JA, et al. Effect of optic nerve sheath fenestration on papilledema of the operated and the contralateral nonoperated eyes in idiopathic intracranial hypertension. Ophthalmology 2011;118:412-4. [Crossref] [PubMed]
- Nithyanandam S, Manayath GJ, Battu RR. Optic nerve sheath decompression for visual loss in intracranial hypertension: report from a tertiary care center in South India. Indian J Ophthalmol 2008;56:115-20. [Crossref] [PubMed]
- Gupta AK, Gupta A, Kumar S, et al. Endoscopic endonasal management of pseudotumor cerebri: Is it effective? Laryngoscope 2007;117:1138-42. [Crossref] [PubMed]
- Chandrasekaran S, McCluskey P, Minassian D, et al. Visual outcomes for optic nerve sheath fenestration in pseudotumour cerebri and related conditions. Clin Exp Ophthalmol 2006;34:661-5. [Crossref] [PubMed]
- Knapp C, Sampath R. Optic nerve sheath fenestration: A five year audit. Neuro-Ophthalmology 2005;29:173-7. [Crossref]
- Thuente DD, Buckley EG. Pediatric optic nerve sheath decompression. Ophthalmology 2005;112:724-7. [Crossref] [PubMed]
- Banta JT, Farris BK. Pseudotumor cerebri and optic nerve sheath decompression. Ophthalmology 2000;107:1907-12. [Crossref] [PubMed]
- Goh KY, Schatz NJ, Glaser JS. Optic nerve sheath fenestration for pseudotumor cerebri. J Neuroophthalmol 1997;17:86-91. [Crossref] [PubMed]
- Acheson JF, Green WT, Sanders MD. Optic nerve sheath decompression for the treatment of visual failure in chronic raised intracranial pressure. J Neurol Neurosurg Psychiatry 1994;57:1426-9. [Crossref] [PubMed]
- Kelman SE, Heaps R, Wolf A, et al. Optic-Nerve Decompression Surgery Improves Visual Function in Patients with Pseudotumor Cerebri. Neurosurgery 1992;30:391-5. [Crossref] [PubMed]
- Kelman SE, Sergott RC, Cioffi GA, et al. Modified Optic-Nerve Decompression in Patients with Functioning Lumboperitoneal Shunts and Progressive Visual-Loss. Ophthalmology 1991;98:1449-53. [Crossref] [PubMed]
- Spoor TC, Ramocki JM, Madion MP, et al. Treatment of pseudotumor cerebri by primary and secondary optic nerve sheath decompression. Am J Ophthalmol 1991;112:177-85. [Crossref] [PubMed]
- Sergott RC, Savino PJ, Bosley TM. Modified optic nerve sheath decompression provides long-term visual improvement for pseudotumor cerebri. Arch Ophthalmol 1988;106:1384-90. [Crossref] [PubMed]
- Corbett JJ, Nerad JA, Tse DT, et al. Results of optic nerve sheath fenestration for pseudotumor cerebri. The lateral orbitotomy approach. Arch Ophthalmol 1988;106:1391-7. [Crossref] [PubMed]
- Brourman ND, Spoor TC, Ramocki JM. Optic nerve sheath decompression for pseudotumor cerebri. Arch Ophthalmol 1988;106:1378-83. [Crossref] [PubMed]
- Smith KA, Peterson JC, Arnold PM, et al. A case series of dural venous sinus stenting in idiopathic intracranial hypertension: association of outcomes with optical coherence tomography. Int J Neurosci 2017;127:145-53. [Crossref] [PubMed]
- Satti SR, Leishangthem L, Spiotta A, et al. Dural venous sinus stenting for medically and surgically refractory idiopathic intracranial hypertension. Interv Neuroradiol 2017;23:186-93. [Crossref] [PubMed]
- Liu KC, Starke RM, Durst CR, et al. Venous sinus stenting for reduction of intracranial pressure in IIH: a prospective pilot study. J Neurosurg 2017;127:1126-33. [Crossref] [PubMed]
- Teleb MS, Cziep ME, Issa M, et al. Stenting and angioplasty for idiopathic intracranial hypertension: a case series with clinical, angiographic, ophthalmological, complication, and pressure reporting. J Neuroimaging 2015;25:72-80. [Crossref] [PubMed]
- Degnan A, Levy L. Pseudotumor cerebri: brief review of clinical syndrome and imaging findings. AJNR Am J Neuroradiol 2011;32:1986-93. [Crossref] [PubMed]
- Jackson IJ, Snodgrass SR. Peritoneal shunts in the treatment of hydrocephalus and increased intracranial pressure: a 4-year survey of 62 patients. J Neurosurg 1955;12:216-22. [Crossref] [PubMed]
- Vander Ark GD, Kempe LG, Smith DR. Pseudotumor cerebri treated with Lumbar-peritoneal shunt. JAMA 1971;217:1832-4. [Crossref] [PubMed]
- Ahmed R, Zmudzki F, Parker G, et al. Transverse sinus stenting for pseudotumor cerebri: a cost comparison with CSF shunting. AJNR Am J Neuroradiol 2014;35:952-8. [Crossref] [PubMed]
- Owler BK, Parker G, Halmagyi GM, et al. Pseudotumor cerebri syndrome: venous sinus obstruction and its treatment with stent placement. J Neurosurg 2003;98:1045-55. [Crossref] [PubMed]
Cite this article as: Scherman DB, Dmytriw AA, Nguyen GT, Nguyen NT, Tchantchaleishvili N, Maingard J, Asadi H, Brooks M, Griessenauer C, Ogilvy C, Thomas AJ, Moore JM, Phan K. Shunting, optic nerve sheath fenestration and dural venous stenting for medically refractory idiopathic intracranial hypertension: systematic review and meta-analysis. Ann Eye Sci 2018;3:26.


